Features
-
Analytical Services | APIs | Bioanalytical Services | Bioassay Development | Biosimilars | Clinical Trials | Drug Development | GMPs/GCPs | Methods Development | Process Development | R&D | Toxicology

Newsmakers: John Robson
Covance’s early development VP talks about CMC, Sanofi, and moreGil Y. Roth 11.14.11
-
Analytical Services | APIs | Bioanalytical Services | Bioassay Development | Chemistry | Drug Development | Excipients | Methods Development | Process Development | Toxicology

Trace Impurity Identification
A combination of spectrometric and spectroscopic techniquesDennis J. Milanowski and Ulla Mocek 11.14.11
-
Analytical Services | Bioanalytical Services | Bioassay Development | Capsules | Drug Development | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Information Technology | Inspections | Lyophilization | Methods Development | Process Development | Process Validation | QA/QC | Risk Management | Toxicology | Validation
Quality by Design: Product & Process Development
Product & Process DevelopmentDr. Siegfried Schmitt 11.14.11
-
Analytical Services | Bioanalytical Services | Bioassay Development | Biologics, Proteins, Vaccines | Biosimilars | Capsules | Excipients | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development | Process Validation | Toxicology | Validation
Virus Safety of Biopharmaceuticals
Absence of evidence is not evidence of absenceHazel Aranha, Ph.D. 11.14.11
-
Clinical Trials | GMPs/GCPs | Information Technology

Collaborative Document Exchange
Listening to the investigator can yield competitive advantagePaul Cofini 11.14.11
-
Biologics, Proteins, Vaccines | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Methods Development | Process Development | QA/QC | Scale-up/Technology Transfer | Validation

Top 10 Mistakes Clients Make with their CMOs
How to improve relations with bio-CMOsEric S. Langer 11.14.11
-
Biosimilars | Drug Development | Regulatory Affairs
Regulatory Leaders
Demand for A-level clinical development and regulatory affairs leadership remains strongVictor Kleinman 10.17.11
-
Analytical Services | Bioanalytical Services | Bioassay Development | Biologics, Proteins, Vaccines | Capsules | Clinical Trial Materials | Clinical Trials | Cold Chain Management | Cytotoxics and High Potency Manufacturing | Drug Delivery | Extractables and Leachables | Facilities | Fill/Finish | Formulation Development | GMPs/GCPs | Logistics | Lyophilization | Process Development | R&D | Solid Dosage/Semi-solids | Supply Chain | Toxicology

-
Capsules | Clinical Trial Materials | Clinical Trials | Drug Development | Drug Discovery | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Logistics | Lyophilization | Methods Development | Process Development | QA/QC | R&D | Validation

Lean Six Sigma and Supplier Relationships
Applying the principles to drive process improvementEsther Sadler-Williams 10.11.11
-
Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Methods Development | Process Development | QA/QC | Supply Chain | Validation
Contract Manufacturing Partnerships
Identifying the “Best Mix” of the right partnersWesley Ange 10.11.11
-
Analytical Services | Aseptic Processing | Bioanalytical Services | Bioassay Development | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Laboratory Testing | Lyophilization | Methods Development | Microbiology | Process Development | QA/QC | Toxicology | Validation

Environmental Monitoring
Make the invisible environment visibleDr. Martin Cockcroft, CBiol.,u2008MSB 10.11.11
-
APIs | Excipients | Extractables and Leachables | Facilities | GMPs/GCPs | Inspections | Logistics | Methods Development | Process Development | QA/QC | Regulatory Affairs | Serialization | Supply Chain | Validation

Supply Chain Focus: Rx-360 Pilot Program
Will audit standards help improve processes?Tom Beil 10.11.11
-
APIs | Capsules | Excipients | Formulation Development | Logistics | Serialization | Solid Dosage/Semi-solids | Supply Chain
Supply Chain Focus: Purity Matters
Third-party certification of propylene glycol USP/EP: one answer to supply chain concernsVincent Lacoste 10.11.11
-
Chemistry

Filling the Gap
Overcoming the manufacturing challenges of hyaluronic acid in dermal fillersHans Ole Klingenberg 10.11.11
-
Aseptic Processing | Biologics, Proteins, Vaccines | Drug Delivery | Fill/Finish | Injectables | Parenterals | Validation | Vials
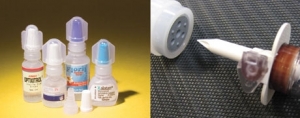
Streamlined Blow-Fill-Seal Insertion
Technology increases flexibility and safety in aseptic packagingAndrew W. Goll 10.11.11
-
Analytical Services | Bioanalytical Services | Bioassay Development | Laboratory Testing | Microbiology | Toxicology | Validation

Rapid Product Release Testing
Shortening production cycle time with rapid microbial screening offers significant benefits for providers and sponsorsCindy Lieberman 10.11.11
-
Clinical Trials | GMPs/GCPs | Information Technology
The Struggle with Clinical Study Budgeting
Industry survey reports problems with accuracy and efficiencyAndrew Grygiel 10.11.11
-
Bioassay Development | Clinical Trials | Cold Chain Management | Drug Development | Drug Discovery | GMPs/GCPs | Inspections | Logistics | Methods Development | Process Development | Regulatory Affairs | Serialization | Toxicology

Maintaining the Focus for Neglected Diseases
Product development partnerships seek to bridge the healthcare gapFaiz Kermani 10.11.11
-

The China Challenge
Balance short-term profits and long-term competitive advantageAndrew Badrot 10.11.11
-
APIs | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Injectables | Logistics | Lyophilization | Process Development | Scale-up/Technology Transfer | Serialization | Supply Chain

Newsmakers: Paul Duffy
A new operating unit helps Pfizer manage 500 external suppliersGil Y. Roth 10.11.11













