Search Results for 'Biomanufacturing'
-
APIs | Biologics, Proteins, Vaccines | Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Lyophilization | Process Development | Validation
Novasep to Manufacture Mydicar API
To provide custom biomanufacturing for commercial production02.17.15
-
Biologics, Proteins, Vaccines | Biosimilars | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development
7 Top Trends in Biomanufacturing Outsourcing
Keep your eye on these trends in 2015.Eric S. Langer and Jean-Claude Lupis, BioPlan Associates 01.29.15
-
Bioassay Development | Biologics, Proteins, Vaccines | Breaking News | Capsules | Collaborations & Alliances | Drug Development | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Methods Development | Process Development | Toxicology
Catalent, Valerion in Biologics Development Pact
Catalent to provide cell line engineering, process development, and manufacturing12.02.14
-
Biologics, Proteins, Vaccines | Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Lyophilization | Process Development
Amgen Opens Next-Gen Bio-Mfg. Facility in Singapore
Incorporates multiple technologies aimed at greater speed and efficiency11.20.14
-
APIs | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Injectables | Inspections | Lyophilization | Process Development | Regulatory Affairs

Newsmakers: CMOs and CDMOs Have a Trade Association to Call Their Own
The new Pharma & Biopharma Outsourcing Association aims to help advance the industry’s regulatory, legislative and business interests.11.13.14
-
Biosimilars | Process Development
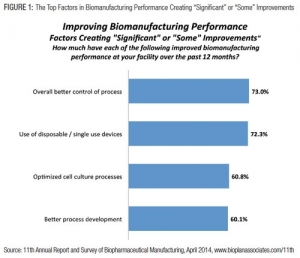
Biopharm Executives Point to Better CMO Performance
Better process control and increased adoption of single-use systems played a part in improving downstream manufacturingEric S. Langer, BioPlan Associates, Inc. 10.14.14
-
APIs | Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Inspections | Lyophilization | Process Development | Regulatory Affairs
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development | Validation
EMD Millipore Expands Biomanufacturing Center in Japan
Training center to aid biopharma companies with manufacturing challenges08.27.14
-

-
Biologics, Proteins, Vaccines | Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Lyophilization | Process Development
Repligen Acquires Refine’s Bioprocessing Business
Gains Alternating Tangential Flow (ATF) System06.03.14
-
Breaking News | cGMP Manufacture | Clinical Trials | Extractables and Leachables | Industry News | Lyophilization | Process Development | Supply Chain | Toxicology | Validation | Vials
-
Biosimilars | Clinical Trials | Drug Delivery | Fill/Finish
Alvotech and Finesse Enter Bio-Manufacturing Pact
Catalent, CiRA in regenerative therapy pact04.03.14
-
Breaking News
-
Biologics, Proteins, Vaccines | Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Lyophilization | Process Development
Catalent Ups Stake in Redwood Bioscience
SMARTag ADC Technology achieves key milestoneKristin Brooks 03.24.14
-
Bio News | Biosimilars | Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development
Alvotech and Finesse Enter Bio-Mfg. Pact
Will leverage SmartFactory single-use automation platformKristin Brooks 03.14.14
-
Biosimilars | cGMP Manufacture | Toxicology
-
Breaking News | Capsules | cGMP Manufacture | Clinical Trial Materials | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Lyophilization | Process Development
Gallus, Symphogen in Clinical Manufacturing Pact
To manufacture Symphogen’s antibody mixture product candidatesKristin Brooks 02.26.14
-
Breaking News | Promotions & Moves
IPS Names Process Engineering Director
Schuster brings 25 years of global experienceKristin Brooks 01.23.14
-
Breaking News | Promotions & Moves
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Capsules | Clinical Trials | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Lyophilization | Process Development
-
Biologics, Proteins, Vaccines | Biosimilars | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development
CMOs Demanding More Innovations
Chromatography and single-use equipment top listEric Langer , BioPlan Associates 11.13.13
-
Bio News | Bioassay Development | Biologics, Proteins, Vaccines | Breaking News | Capsules | cGMP Manufacture | Drug Development | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Inspections | Lyophilization | Methods Development | Process Development | R&D | Regulatory Affairs | Toxicology
-
Breaking News | Promotions & Moves
-
Biosimilars | Validation
-

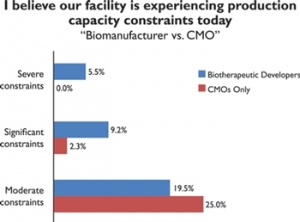
Transforming Bigfoot Into Speed Racer
The facility of the future movement in biologics manufacturingJeff Odum, IPS 09.05.13
-
Breaking News | Clinical Trial Materials | Industry News | Process Development
Gallus, Genzyme in Clinical Supply Pact
To provide process and method development, scale-up, clinical supply for rare disease therapy08.01.13
-
Breaking News | Capsules | Clinical Trial Materials | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Lyophilization | Process Development
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | Formulation Development | GMPs/GCPs | Industry News | Lyophilization | Microbiology | Process Development
-
Breaking News | Promotions & Moves
IPS Appoints Compliance Director for Latin Americas
DeSeabra brings 20 years of leadership experience04.10.13
-
Clinical Trials | Fill/Finish | Process Development | Validation

Global Changes in Biomanufacturing
A retrospective analysis of CMO TrendsEric Langer, BioPlan Associates, Inc. 04.03.13
-
Biosimilars | Clinical Trials | Drug Delivery | Drug Development | Process Development | Validation

Bio-CMO Industry Trends
The market continues its upswingWilliam Downey and Jennifer Hartigan, HighTech Business Decisions 04.03.13
-
Aseptic Processing | Biosimilars | Clinical Trial Materials | Clinical Trials | Fill/Finish | Lyophilization | Process Development | Validation | Vials
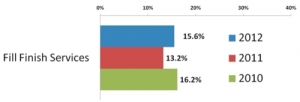
Fill-Finish Innovation
Showing Sparks of LifeRonald A. Rader and Eric S. Langer, BioPlan Associates, Inc. 03.13.13
-
Biosimilars | Breaking News | Collaborations & Alliances
Viropro, Oncobiologics in Biosimilars Pact
Aims to commercialize six products in emerging markets02.26.13
-
Breaking News | Drug Development | Drug Discovery | Industry News | Methods Development
Amgen Licenses DSM’s XD Cell Culture Patents
Technology is applicable to multiple biological products01.15.13
-
Bioassay Development | Clinical Trial Materials | Clinical Trials | Drug Development | Drug Discovery | Laboratory Testing | Logistics | Methods Development | Process Development | Toxicology | Validation

Clinical Testing Technology
Applying flow cytometry to clinical trial samplesKevin Maskell, Merck Millipore 11.09.12
-
Biosimilars

Biosimilars: The Race to Market Continues…
Going for the GoldFiona Greer, SGS Life Science Services 10.09.12
-
Biologics, Proteins, Vaccines | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development | Scale-up/Technology Transfer
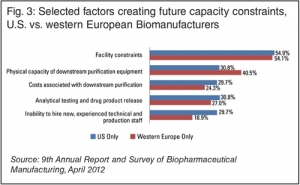
Future Bio-Capacity Constraints
Top 10 factors from a new surveyEric S. Langer, BioPlan Associates, Inc. 09.06.12
-
Analytical Services | Bioanalytical Services | Bioassay Development | Biologics, Proteins, Vaccines | Biosimilars | Process Validation | Toxicology
Continuous Flow Manufacturing
Implications on downstream biologics processingAloke Das, Beroe Inc. 09.06.12
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Capsules | Clinical Trial Materials | Clinical Trials | Drug Discovery | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Laboratory Testing | Lyophilization | Process Development
WuXi PharmaTech To Make HIV MAb for TaiMed
Biomanufacturing deal covers global Phase II-III trialsGil Roth 08.28.12
-
Analytical Services | Bio News | Bioanalytical Services | Bioassay Development | Biologics, Proteins, Vaccines | Breaking News | Capsules | Clinical Trials | Drug Development | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Methods Development | Process Development | R&D | Toxicology
Fujifilm Diosynth To Expand Bio-Capabilities
Will boost cGMP mammalian cell culture at BillinghamGil Roth 06.19.12
-
Bio News | Bioassay Development | Biologics, Proteins, Vaccines | Breaking News | Capsules | Drug Development | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Methods Development | Process Development | R&D | Scale-up/Technology Transfer | Toxicology
-
Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Industry News | Lyophilization | Process Development
-
Analytical Services | Aseptic Processing | Bioanalytical Services | Bioassay Development | Biologics, Proteins, Vaccines | Biosimilars | Capsules | Clinical Trial Materials | Clinical Trials | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development | Process Validation | Scale-up/Technology Transfer | Toxicology

Bio-CMO Industry Trends
Recent initiatives in biopharma contract manufacturingWilliam Downey, HighTech Business Decisions 05.04.12
-
Analytical Services | Aseptic Processing | Bioanalytical Services | Bioassay Development | Biologics, Proteins, Vaccines | Biosimilars | Capsules | Clinical Trial Materials | Clinical Trials | Drug Development | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Inspections | Logistics | Lyophilization | Methods Development | Process Development | QA/QC | R&D | Regulatory Affairs | Scale-up/Technology Transfer | Serialization | Supply Chain | Toxicology | Validation
Biosimilar Outsourcing Update
FDA’s draft guidance leads to new opportunitiesGil Roth, Contract Pharma 05.04.12
-
Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development
-
Drug Development | Logistics
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Capsules | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development
GE Healthcare To Acquire Xcellerex
Boosts single-use biopharma manufacturing businessGil Roth 03.07.12
-
Analytical Services | Bioanalytical Services | Bioassay Development | Capsules | Cleaning Validation | Extractables and Leachables | Facilities | Fill/Finish | Formulation Development | GMPs/GCPs | Inspections | Laboratory Testing | Lyophilization | Methods Development | Process Development | Process Validation | QA/QC | Solid Dosage/Semi-solids | Toxicology | Validation | Vials
Manufacturing Equipment Trends
Industry perspective from suppliers and manufacturersOnline Exclusives Kristin Brooks, Associate Editor 03.07.12
-
Bio News | Biologics, Proteins, Vaccines | Biosimilars | Breaking News | Capsules | Collaborations & Alliances | Extractables and Leachables | Facilities | Fill/Finish | GMPs/GCPs | Lyophilization | Process Development
GE, M+W in Biosimilar Engineering Pact
To develop biomanufacturing facilities in emerging marketsGil Roth 12.14.11
-
Breaking News | Process Development












