Search Results for 'Development'
-
Breaking News | Clinical Trials
ICON plc Selected by BARDA to Execute Anthrax Vaccine Clinical Trial
BARDA is providing funding to develop and execute a clinical trial that will evaluate the immune response to AV7909.10.10.22
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Collaborations & Alliances | Industry News

Myrtelle and Forge Biologics Enter Manufacturing Partnership
To advance Myrtelle’s novel gene therapy for monogenic hearing loss for Phase 1/2 clinical trials.10.07.22
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Collaborations & Alliances | Facilities | Industry News
BioNTech and Australia’s State of Victoria Form Partnership
Will establish mRNA research center and manufacturing facility.10.07.22
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Industry News

CEPI and Intravacc Partner to Develop Betacoronavirus Vaccine
CEPI will provide seed funding of up to $4.8 million to Intravacc to advance R&D of preventative therapeutic vaccines.10.06.22
-
Biologics, Proteins, Vaccines | Breaking News | cGMP Manufacture | Industry News

Cytiva Acquires CEVEC Pharmaceuticals
Strengthens viral vector manufacturing and cell line development technologies for gene therapies.10.06.22
-
Clinical Trials

DCTs Deliver Measurable Benefits for Sponsors: Global Analysis Findings
Multiple benefits impacting trial timelines and related cost with notable reductions in protocol deviations rate, recruitment times, and first patient in.Online Exclusives Bhausaheb Patil, Head of Business Operations, Decentralized Clinical Trials, IQVIA 10.06.22
-
Biologics, Proteins, Vaccines | Breaking News | cGMP Manufacture | Industry News

Univercells Completes Expansion of Nivelles, Belgium Site
€2 million investment aims to deliver greater support and supply chain security to biomanufacturers.10.05.22
-
Biologics, Proteins, Vaccines | Breaking News | Industry News | Supply Chain

Almac Introduces New Cryogenic Service Solution
Invests $4 million in next generation cold chain solutions at the company’s Durham, NC facility.10.05.22
-
Cold Chain Management | Laboratory Testing | Vials

Meeting the Container Closure Integrity Requirements in the Revised EU Annex 1
This webinar will present how sterile product manufacturers can prepare themselves for the new regulations.10.05.22
-
Breaking News | Clinical Trials | CRO News | Drug Development

WCG Publishes Safety Reference Model Operational Guide
Was developed in response to the critical need to harmonize safety reporting processes from regulators and across sponsors and CROs.10.04.22
-
Breaking News | Facilities | R&D

Eisai Completes New Injection/Research Building in Japan
EMITS will be Eisai's global base for formulation and modality research.10.04.22
-
APIs | Biologics, Proteins, Vaccines | Breaking News | cGMP Manufacture | Industry News

MilliporeSigma Opens Commercial Facility for New CTDMO Services
The 2,700-square-meter site in Martillac, France will provide global supply of commercial drug substances.10.04.22
-
Biologics, Proteins, Vaccines | Breaking News | CRO News | Drug Discovery | Laboratory Testing

BioIVT, Charles River Labs Enter Global eCommerce Collaboration
Charles River Avian products for virus research are now available through the BioIVT Digital Portal.10.04.22
-
Breaking News | Collaborations & Alliances | Drug Discovery

Astellas Pharma and Pantherna Therapeutics Enter New Technology Evaluation Agreement
Pantherna's mRNA platform & Astellas' drug discovery capabilities will be combined to generate mRNA-based regenerative medicine programs.10.04.22
-
Drug Delivery
The Future of Drug Delivery Technology
Drug delivery technologies represent the application of science to exploit physical mechanisms in biological systems.Ben Locwin, Contributing Editor 10.03.22
-
Analytical Services | Laboratory Testing

Quality and Profitability
Not an either/or proposition.Emil W. Ciurczak, DoraMaxx Consulting 10.03.22
-
Drug Development | Drug Discovery | R&D

CEO Spotlight: Jonathan Hunt
Syngene International chief executive explains how the traditional cost arbitrage model is evolving to a more rewarding intellectual arbitrage.S. Harachand, Contributing Writer 10.03.22
-
Clinical Trials | Information Technology

Data Integrity
FDA guidance on real-world data: how it affects clinical trial design and impacts patients.Karen Ooms, Joint Chief Operating Officer, Quanticate 10.03.22
-
Drug Delivery | Drug Development

Early Phase to Drug Product
Advantages of an integrated development model.Stephanie Sastre and Mark Cappucci, Lonza Small Molecules 10.03.22
-
Analytical Services | Facilities | Laboratory Testing

Azelis Mexico’s New Innovation Center
The 10,000 square-foot facility is designed to facilitate more collaboration with customers in the pharma industry.Tom Branna, Editorial Director 10.03.22
-
Biologics, Proteins, Vaccines

Trends Toward Outsourcing Buffer Manufacturing
As the cell and gene therapy market continues to grow, large and small biopharmas are increasingly relying on third parties to source their buffers.Kate Buggle, Strategic Business Development Director, Actylis 10.03.22
-
Analytical Services | Reader Showcase

Thermo Fisher Scientific Introduces Orbitrap Ascend Tribrid
Designed to enable improved multiplexed proteomics and native protein characterization.10.03.22
-
Drug Development | Drug Discovery | Preclinical Outsourcing

Streamlining Early Phase Discovery and Development
Early phase drug development is often defined by speed, but planning is essential to avoid added costs and delays in the later stages of a molecule’s journey.Mat Minardi and John Mykytiuk, Sterling Pharma Solutions 10.03.22
-
Formulation Development | Reader Showcase

Asahi Kasei Introduces MOTIV Buffer Management
MOTIV is designed to keep pharmaceutical production moving efficiently and precisely.10.03.22
-
cGMP Manufacture | Drug Development | Fill/Finish | R&D

Contract Pharma Sits Down with Patrick Haffey of Selkirk Pharma
CEO of Fill/Finish CMO shares insight on the future of the Pharmaceutical industry.Videos
-
Breaking News | Drug Development | Industry News

Novo Nordisk, Ventus Enter Licensing Deal with Potential Value of $700M
To develop and commercialize peripherally-restricted NLRP3 inhibitors to target a broad range of diseases, including NASH and chronic kidney disease.09.30.22
-
Breaking News | Clinical Trials | CRO News

Emmes Acquires Clinical Edge
Expands ophthalmic certification and training services for clinical trials across all phases of development.09.30.22
-
Biologics, Proteins, Vaccines | Breaking News | Collaborations & Alliances | R&D
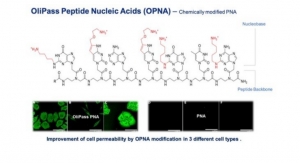
Vanda Pharmaceuticals, OliPass Enter ASO R&D Alliance
Aims to jointly develop a set of antisense oligonucleotide (ASO) molecules based on OliPass' modified peptide nucleic acids.09.30.22
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Industry News

Center for Breakthrough Medicines and jCyte Ink Manufacturing Deal
Multi-year deal to focus on phase 3 clinical trial supplies and commercial manufacturing.09.29.22
-
Breaking News | Clinical Trial Materials | Collaborations & Alliances | Drug Development | Industry News
Transpire Bio Inks Second Agreement with Recipharm
Will develop two additional inhaled medicines.09.29.22
-
Breaking News | Drug Delivery | Facilities | Industry News

Vectura’s Inhalation Center of Excellence Granted Planning Permission
$62 million building to be completed in 2025.09.27.22
-
Drug Development | Laboratory Testing | Parenterals

Release & Stability Testing Requirements for Parenteral Drug Products
This presentation will provide a brief overview of the parenteral drug products release testing as well as the ICH guidelines for stability testing.09.26.22
-
Biologics, Proteins, Vaccines

Pfizer & BioNTech Request EUA of Omicron BA.4/BA.5-Adapted Bivalent Vaccine Booster in Children
The request is supported by safety and immunogenicity data from multiple studies.09.26.22
-
Breaking News | CRO News | Drug Discovery | Industry News | Laboratory Testing | R&D

BioIVT Acquires Fidelis Research
Extends capabilities to deliver fresh biospecimens and produce cell products for drug and diagnostic development.09.26.22
-
Analytical Services | Breaking News | Facilities

Frontage Laboratories Opens Custom-Built Laboratory in Hayward, CA
New facility will support the local Bay Area biotech and pharmaceutical community.09.23.22
-
Breaking News | R&D

Abenza Upgrades Research & Development Capabilities in Cambridge
Invests in B cell discovery technologies and appoints Jamie Campbell as Vice President of Research & Development.09.23.22
-
Biologics, Proteins, Vaccines | Breaking News | cGMP Manufacture | Industry News

FUJIFILM Diosynth Biotechnologies Expands Microbial Mfg. Facility in Billingham, UK
Large-scale facility will include more than 20,000 sq. ft. of modular clean room space and will increase microbial production by 70%.09.23.22
-
Breaking News

MedPharm to Support Mosanna Therapeutics in the Development of MOS-118
New drug aims to tackle Metabolic Obstructive Sleep Apnea.09.21.22
-
Breaking News | Clinical Trials

Dr. Vince Clinical Research Opens Clinical Pharmacology Unit and HQ in Kansas
The custom-built, green-inspired research complex was designed and engineered to be a premier destination for Phase I clinical studies.09.21.22
-
Biologics, Proteins, Vaccines | Breaking News | Collaborations & Alliances

Rokote Laboratories Licenses Cevec’s Proprietary CAP Ad Technology
Gains non-exclusive rights to use the CAP Ad platform for the manufacturing of adeno-based vaccines for prophylactic and therapeutic use in infectious diseases.09.21.22
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Facilities | Industry News

Cellistic Acquires Celyad Oncology’s Manufacturing BU
Will invest substantial capital into the newly acquired 11,000 square foot facility.09.20.22
-
Breaking News | Collaborations & Alliances | Drug Development

CytoReason Extends multi-Year Partnership with Pfizer
Will continue to provide its artificial intelligence technology for Pfizer’s drug development programs.09.20.22
-
Biologics, Proteins, Vaccines | Breaking News | Collaborations & Alliances

Valneva and IDT Biologika Terminate Covid-19 Collaboration
In light of the reduced European Commission order, Valneva has suspended manufacturing of the vaccine.09.19.22
-
Clinical Trials | Formulation Development | Process Development | Regulatory Affairs | Solid Dosage/Semi-solids

8 Trends in Contract Manufacturing
As both a CDMO and a manager of CMOs, Avéma Pharma Solutions has seen 8 trends emerge in Contract Manufacturing.
-
Breaking News | Drug Development | Industry News | Toxicology

BioIVT Acquires XenoTech
XenoTech specializes in ADME, DMPK and DDI testing of potential drug candidates.09.15.22
-
Breaking News | Clinical Trials | Collaborations & Alliances

Thermo Fisher’s PPD Clinical Research Business to Assist NCI’s Cancer Trials Support Unit
Project will be funded with federal funds from the National Cancer Institute, National Institutes of Health and Department of Health and Human Services.09.15.22
-
Breaking News | Facilities | Formulation Development | Industry News

Bora Expands CDMO Capabilities with Acquisition of TWi Pharmaceuticals
Adds two manufacturing facilities and strengthens service offerings in formulation development, sterile ophthalmic, and niche manufacturing technologies.09.14.22
-
Breaking News | Fill/Finish | Industry News | Lyophilization | Parenterals | R&D | Toxicology

Berkshire Sterile Manufacturing Expands Services
Capabilities now include R&D and TOX lot formulation and filling.09.14.22
-
Analytical Services | Bioanalytical Services | Laboratory Testing | QA/QC

Considerations When Outsourcing Stability Testing
Stability studies are a critical element of drug development and a key commitment in maintaining product on the market.Paul Mason, Lachman Consultants 09.13.22
-
Biologics, Proteins, Vaccines | Information Technology | R&D













