Search Results for 'Outsourcing'
-
Chemistry | Clinical Trials | Drug Discovery | Industry News | Laboratory Testing | Preclinical Outsourcing
Novasep Opens Expanded U.S. Kilo Lab
Begins production of batches of synthetic molecules for early stage development06.02.16
-
Breaking News | CRO News | Drug Development | Promotions & Moves
ClinTec Expands Capabilities
Opens offices in Spain and Portugal; appoints regional manager05.23.16
-
Breaking News | Drug Development | Logistics | Promotions & Moves
PCI Appoints Int’l Biz Development Manager
Pieter-Jan Vos will be responsible for operations in Germany, The Netherlands and Switzerland05.13.16
-

2016 Annual Outsourcing Survey
Herein are the results of our 2016 Annual Outsourcing Survey05.11.16
-
Clinical Trials | Drug Development | Information Technology | R&D
CROs and Today's R&D Landscape
John Lewis of ACRO discusses opportunities and challenges, global markets, and the future CROOnline Exclusives Kristin Brooks, Contract Pharma 05.11.16
-
APIs | Chemistry | Drug Delivery | Drug Development | Drug Discovery | Formulation Development | Industry News | Preclinical Outsourcing
-
Biosimilars | Risk Management
India Revises Biosimilars Guidelines
Stricter quality, safety norms on cardsS. Harachand, Contributing Editor 05.05.16
-
Capsules | Validation
Sometimes You Need to Spend to Save
Exploring the importance of investing to develop new analytical methodsEmil W. Ciurczak, DoraMaxx Consulting 05.05.16
-
APIs

2016 Annual Outsourcing Survey
Herein are the results of our 2016 Annual Outsourcing SurveyTim Wright, Editor 05.05.16
-
APIs | Cleaning Validation | Clinical Trials | Drug Development | Drug Discovery | Risk Management | Toxicology | Validation

Setting Health-Based Exposure Limits to Support Pharmaceutical Development and Manufacturing
Health-based exposure limits are needed to support risk assessmentsB.D. Naumann, P.J. Nigro, U. Bruen, L. Parola, D. Cragin and A. Schatz, Merck & Co., Inc. and Ashland, Inc. 05.05.16
-
Clinical Trials | Drug Development | Regulatory Affairs | Toxicology

CRO Industry Update
Harnessing Data in the Age of ComplexityKristin Brooks, Contract Pharma 05.05.16
-
Capsules | Clinical Trials | Excipients | Formulation Development | Serialization

The Evolution of the ‘One-Stop Shop’ in Clinical Supplies Sourcing
What does a one-stop shop really mean and is it always best for the client?Paul Skultety, Alex McClung, Ted Koontz and Damian Gant, Xcelience, a division of Capsugel Dosage Form Solutions 05.05.16
-
Biosimilars | Clinical Trials | Process Development

Biosimilars Outsourcing and Capacity
CMOs to play important but uncertain role in biosimilars manufacturingRonald A. Rader and Eric S. Langer, BioPlan Associates 05.05.16
-
Clinical Trials | Drug Discovery | Promotions & Moves
SYMBIOSIS Expands North American BD Team
Scullion to aid commercial interests and client relationships in U.S.05.02.16
-
Breaking News | Drug Development | Drug Discovery | Preclinical Outsourcing | R&D | Reader Showcase
Recursion Pharma In Research Pact With Sanofi Genzyme
Will use its drug repurposing platform to identify new uses for Sanofi’s clinical stage molecules04.25.16
-
Analytical Services | Formulation Development
Catalent Names Development & Analytical Services VP
Langa responsible for growing the early phase development business for small molecule compounds04.21.16
-
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Drug Development | Drug Discovery | Industry News | Preclinical Outsourcing
-
APIs | cGMP Manufacture | Chemistry | Drug Discovery | Process Development | Supply Chain
API Sourcing Trends
Drug product complexities, quality and regulatory hurdles drive market shiftOnline Exclusives Kristin Brooks, Contract Pharma 04.11.16
-
Ajinomoto Althea Expands Its Capacity of Fill Finish Manufacturing with Addition of Second Shift
...
-
cGMP Manufacture | Facilities | Lyophilization | Scale-up/Technology Transfer | Solid Dosage/Creams/Ointments
Flexible Manufacturing Strategies
Michael Lehmann of Patheon discusses solutions for pharmaceutical forecasting errorsOnline Exclusives Kristin Brooks, Contract Pharma 04.04.16
-
Breaking News | cGMP Manufacture | Industry News
Adamis Pharma Acquires US Compounding
US Compounding will become a wholly-owned subsidiary of Adamis03.29.16
-
APIs | Drug Development | Formulation Development | Logistics | R&D | Solid Dosage/Creams/Ointments | Supply Chain
DCAT Week ’16 Session Highlights
API and Dose CMO Market Trends and Supply StrategiesOnline Exclusives Kristin Brooks, Contract Pharma 03.24.16
-
Analytical Services | Bioassay Development | Breaking News | Drug Discovery | Industry News | Preclinical Outsourcing
Agilent, Eurofins to Expand SureVector Cloning System
To offer custom components and services for next-gen cloning kits03.11.16
-
Fill/Finish | Process Development | Validation
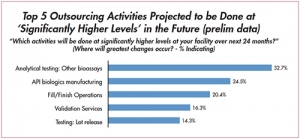
Taking Stock of Biopharma’s Outsourcing Markets
Outsourcing of testing services on the riseEric S. Langer, BioPlan Associates 03.09.16
-
Chemistry | Excipients | Supply Chain | Validation
Outsourcing Manufacturing Really Stretches the Supply Chain
Potential interference from supply chain differences is problematicEmil W. Ciurczak, DoraMaxx Consulting 03.09.16
-
Inspections | Risk Management | Supply Chain
Quality Metrics: What Does it Really Mean?
Background, thoughts and the current industry dialogue on FDA’s quality metrics initiativeDr. Mike Long, ValSource 03.09.16
-
APIs | Capsules | Chemistry | Drug Delivery | Excipients | Formulation Development | Serialization | Supply Chain

Solid Dosage Outsourcing Trends
The market continues to evolve as blockbusters are replaced by more targeted treatments that have unique requirements.Tim Wright, Editor 03.09.16
-
Chemistry | Process Development | QA/QC | Supply Chain | Validation
Contract Manufacturing: Setting Standards in the Supplement Industry
Ensuring nutraceuticals are produced safely and according to strict quality requirements is paramount in the current climate of regulatory scrutiny.Sean Moloughney, Nutraceuticals World 03.09.16
-
APIs | Aseptic Processing | Biosimilars | Fill/Finish | Process Development | Supply Chain

Single-use Technology Integral to Advancing Biomanufacturing
Innovative process technologies are a necessity for today’s biopharmaceutical contract manufacturersNigel Walker, Thatu2019s Nice LLC / Nice Insight 03.09.16
-
APIs | Biosimilars | Drug Delivery | Drug Development | Fill/Finish | Formulation Development | Lyophilization | Parenterals | Supply Chain

Parenteral Outsourcing Trends
Contract Pharma asked a roundtable of business leaders from Symbiosis, Grifols, Althea, LSNE and Baxter to discuss key trends in the parenteral dosage development and manufacturing marketTim Wright, Editor 03.09.16
-
APIs | Chemistry | Clinical Trials | Drug Delivery | Drug Development | Industry News | Injectables | Process Development

Consolidation in Outsourcing
Reviewing 2015’s M&A activityMichael A. Martorelli, Fairmount Partners 03.09.16
-
Capsules | Chemistry | Extractables and Leachables | Fill/Finish | Formulation Development | Supply Chain
Newsmakers: Hanns-Christian Mahler
Head of drug product services talks about Lonza’s new capabilitiesTim Wright, Editor 03.09.16
-
Clinical Trials | Drug Development | Drug Discovery | Industry News | Laboratory Testing | Preclinical Outsourcing | Toxicology
Crown Bioscience Acquires PreClinOmics
Expands cardiovascular and metabolic disease portfolio03.08.16
-
Breaking News | cGMP Manufacture | Clinical Trial Materials | Collaborations & Alliances | Drug Development | Preclinical Outsourcing | Process Development
JHL Biotech and Affinita Biotech Team Up
Partner in the development and manufacturing of oncology monoclonal antibodies03.01.16
-
-
-
Analytical Services | Breaking News | Facilities | Industry News | Laboratory Testing
-
Drug Delivery | Industry News | Serialization | Supply Chain
PBOA Adds New Members and Trustees
3M Drug Delivery Systems, Ei Solutionworks, IDT Biologika join CMO/CDMO association02.16.16
-
Analytical Services | Breaking News | Formulation Development | Promotions & Moves
Langa Joins Catalent as VP of Development & Analytical Services
Previously served as Halo’s VP of sales and marketing02.15.16
-
Analytical Services | Drug Development | Drug Discovery | Financial Reports | Laboratory Testing | Preclinical Outsourcing
-
Clinical Trials | Drug Development | Financial Reports | Laboratory Testing | Preclinical Outsourcing
Financial Report: Charles River Labs
Manufacturing Support segment drives growth in the quarter02.10.16
-
Bio News | Breaking News | cGMP Manufacture | Clinical Trial Materials | Facilities | Industry News
Transgene and ABL Strike Purchase Deal
Entails acquisition by ABL Europe of Transgene’s production asset02.04.16
-
Breaking News | Drug Development | Extractables and Leachables | Formulation Development | Industry News | Parenterals | Process Development
Lonza to Offer Drug Product Development Services
Capabilities will focus on parenteral dosage forms for biologics, drug conjugates, peptides and small molecules02.02.16
-
Drug Discovery | Industry News | Preclinical Outsourcing
MD Biosciences Expands Preclinical Services
Launches new suite of in vivo and in vitro cancer models01.28.16
-
Biosimilars | Clinical Trials
-
Analytical Services | Clinical Trials | Laboratory Testing | Supply Chain
Analytical Instruments are On-Board with Outsourcing
Newer process instruments are finally being designed with mobility in mindEmil W. Ciurczak, DoraMaxx Consulting 01.28.16
-
Clinical Trials | Drug Development | R&D

Optimizing Collaboration Effectiveness in Alliance Partnerships
...Mary Jo Lamberti, Phil Birch, Ranjana Chakravarthy, Ken Getz 01.28.16
-
APIs | Inspections

The Western API Industry
Temporary reprieve or lasting recovery?Dr. Enrico T. Polastro, Arthur D. Little 01.28.16
-
APIs | Drug Development | Formulation Development | Industry News | Logistics | Risk Management | Supply Chain
A Look at DCAT Week ‘16
On the pulse of the pharmaceutical manufacturing value chain, DCAT Week ‘16 will deliver top-flight education programs and networking events.Tim Wright, Editor 01.28.16












