07.01.10
#1 - Roche/Genentech
4070 Basel, Switzerland
Tel: (41) 61 688 1111
Fax: (41) 61 691 9391
www.roche.com
| Headcount | 81,507 | (54,973 in bio/pharma) |
| Year Established | 1896 | |
| Bio/pharma Revenues | $36,017 | +8% |
| Royalty Revenues | $1,182 | +7% |
| Total Revenues | $45,304 | +3% |
| Net Income | $7,528 | -25% |
| R&D Budget | $2,800 | +13% |
| 2009 Top Selling Drugs | |||
| Drug | Indication | Sales | (+/-%) |
| Avastin | oncology | $5,747 | +19% |
| MabThera/Rituxan | rheumatoid arthritis | $5,622 | +2% |
| Herceptin | breast cancer | $4,864 | +3% |
| Tamiflu | influenza | $2,956 | +424% |
| Pegasys/Copegus | hepatitis C | $1,529 | +1% |
| CellCept | transplantation | $1,456 | -25% |
| NeoRecormon/Epogin | anemia | $1,441 | -12% |
| Tarceva | lung cancer | $1,204 | +7% |
| Xeloda | oncology | $1,164 | +4% |
| Lucentis | wet age-related macular degeneration | $1,106 | +26% |
| Boniva | osteoporosis | $977 | -5% |
| Xolair | asthma | $573 | +11% |
| Vacyte | virology | $521 | +2% |
Account for 81% of total bio/pharma sales, up from 77% in 2008.
PROFILE
We have a new #1! With Roche’s acquisition of Genentech, which closed in April 2009, Roche supplanted Amgen and moved into the top rank of global biopharma companies. If I listened to critics and scrapped the Biopharma category, Roche would’ve ranked #5 in the overall list, jumping ahead of AZ, Merck and J&J. But Roche took the message of this acquisition seriously enough that it quit PhRMA and joined BIO, so I’m keeping my Pharma and Biopharma lists separate for another year.
Unlike its Basel neighbor Novartis, Roche reports its results in Swiss francs. Fortunately, the average exchange rates in 2009 and 2008 were almost identical, causing few discrepancies when comparing results. (That said, the franc began appreciating against the dollar in 1Q10, so those numbers may skew a little.)
|
The Lowe Down
And now we have to speak of Roche and Genentech as truly one entity. I’ll admit that I was quite sad about that when it happened — and I still wonder how many people the company might have lost had there been anywhere else to get a job within five hundred miles. But reports from the ground are good. So far, anyway, the folks from Basel seem to have left people more or less alone to do their thing. I wouldn’t have bet on that myself, so I have to congratulate the Swiss for their apparent forethought. Basel and the Bay Area have never been in danger of being mistaken for each other, in either scenery or Weltanschauung (sorry, there’s the language creeping in), but let us not to the marriage of true minds admit impediments. Roche actually has a rare amount of breathing space, compared to their competition, since they’re facing no particularly huge patent expirations. They’ve got some really promising stuff late in the clinic, too. What is this? Stability? Didn’t we get rid of that a few years ago? Let’s hope that they make the most of it.—Derek Lowe |
Roche’s Tamiflu posted the biggest jump of any drug in this year’s report, as H1N1 preparations led to $2.9 billion in sales, up from $564 million in 2008. Of course, that number will drop during 2010, but it sure must have helped Roche to gain $2.4 billion in extra revenues during an integration year. In fact, that number happens to offset almost exactly the $2.2 billion in restructuring/integration charges Roche incurred over the course of 2009! (Roche expects to pay another $900 million or so during 2010 in restructuring charges.)
The biggest charges involved stopping construction on a bulk manufacturing site in Vacaville, CA, shutting down manufacturing in Nutley, NJ, where Roche had operations for 80 years (it’ll keep research at the site), and closing an R&D facility in Palo Alto, CA. Roche also evaluated Genentech’s partnerships and collaborations and did some hardcore prioritizing; in the span of one week in December 2009, Roche ended development projects with Seattle Genetics, Actelion and Genmab.
Roche didn’t scuttle all of Genentech’s deals. In August 2009, Roche exercised Genentech’s option to buy Lonza’s cell culture manufacturing site in Singapore for $290 million, with another $70 million in potential milestones. The site has 80,000 liters of fermentation capacity and will be used to make bulk substance for Avastin. Roche is also building a mammalian facility in Singapore, expected to come online next year.
|
Bio Attack
Other drugmakers are frenziedly working to assemble a biologics portfolio half as extensive as that of Roche, but that doesn’t mean Roche’s run is risk-free. Sales of NeoRecormon/Epogin fell 11% in 2009, due mainly to biosimilar competition in Europe. In May 2010, Teva started recruiting a clinical trial for a biosimilar version of Rituxan, Roche’s #2 product. The trial is only testing against rheumatoid arthritis, which accounted for approximately $830 million of Rituxan’s $5.6 billion in 2009 sales, and Rituxan is patent protected in the U.S. through 2018 (although it expires in the EU in 2013). Still, Teva — with partner Lonza, which helped manufacture Rituxan through 2008 — has fired a biosimilar broadside. |
In addition, the CEOs of Roche’s Pharma and Diagnostics division both retired at the end of 2009, as did the head of research. Genentech’s chief operating officer, Pascal Soriot, was named COO of the Pharma division. His role at Genentech (which is now the U.S. face of Roche) was taken by Ian Clark, previously the head of Pharma’s Global Product Strategy unit.
After all this restructuring and shuffling, Roche expects to save more than $800 million in annual costs.
Is that enough to offset Genentech’s loss of independence? Sure, it wasn’t the scrappy little biotech by the bay anymore, but in an era where the largest pharmas keep touting their new “biotech” structures where they devolve accountability and try to promote an entrepreneurial vibe in companies with more than 75,000 employees, it’s odd to see Roche is busily swallowing up its own perfectly fine biopharma.
But modern pharma is as much about managing business processes as it is about drug development. Since Roche actually has a pretty extensive pipeline of new molecules as well as label extensions for some of its blockbusters, it can afford to focus on cost savings and restructuring globally. And, hey, at least Roche isn’t touting the benefits of generics or consumer healthcare!
|
Acquisition News
Target: Medingo Ltd. Price: $160 million plus $40 million in potential milestones Announced: April 2010 What they said: “With this acquisition we will broaden our portfolio of innovative insulin delivery technologies and strengthen our position as a leading player in the diabetes care business.” —Daniel O’Day, COO, Roche Diagnostics |
This isn’t to say that Roche has no weak spots. CellCept went generic in the U.S. in May 2009, leading to a $500 million fall in sales. Roche’s NeoRecormon fell 11% because of pressure from biosimilars (see Bio Attack on next page). Boniva sank after safety worries led insurers to cut access; the approval of Amgen’s Prolia should drop it even further.
Roche suffered a major setback with its investigational type 2 diabetes treatment, taspoglutide. In a Phase III trial of the GLP-1 drug, which Roche is co-developing with Ipsen, a small but significant number of patients — fewer than 1%, according to Roche — had hypersensitive reactions to it. Roche had been hoping to bring the drug to market in 2011 to compete with Novo Nordisk’s Victoza and Lilly/Amylin’s Byetta, but will now delay filing for at least 12 to 18 months. The company projected peak sales of $1.8 billion for the drug, and some analysts called it as high as $3 billion, but now there’s talk that Roche may end up throwing in the towel and giving the compound back to Ipsen. That’s a terrible blow given Roche’s desire to build a non-oncology franchise.
Roche and partner Biogen Idec also got bad news in March 2010 when ocrelizumab trials in RA were suspended due to safety risks. Trials against lupus were suspended earlier due to safety risks, too. Trials are continuing in MS, but in May 2010 the companies elected to end development in RA, the indication that was key to building another multi-billion-dollar biologic (and extending the RA franchise after Rituxan loses patent protection in a few years).
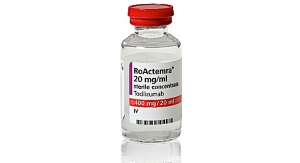
In January 2010, Roche got FDA approval for Actemra, an RA treatment for patients who don’t respond to TNF-alpha inhibitors. The company hopes that further data will allow for earlier prescription of Actemra. Peak sales could top $2 billion, or reports of side effects in Japan, where it was approved earlier, could limit it to a niche product.
Roche suffered failures in several Avastin trials (prostate and gastric cancer, with success in an ovarian cancer trials), but the cancer drug managed to keep rolling on, posting nearly 20% sales growth and becoming Roche’s top seller in 2009. In 1Q10, sales were up another 18% to $1.6 billion. The FDA approved Avastin to treat chronic lymphocytic leukemia in adults in February 2010, and if it manages to add that ovarian cancer indication (or early breast cancer, where it showed good results in a recent trial), Roche will silence (some of) the critics who complained that $47 billion was too high a price for Genentech.
Despite some pipelne setbacks, Roche has several top prospects. The company is already holding three aces — Avastin, Rituxan and Herceptin are all poised to post more than $5 billion in 2010 sales — so the next few years will tell us whether they add enough top-sellers for a full house.
Return to Top Biopharma Report homepage.