YOU SEARCHED BY...
SEARCH BY...
Country
- Canada(10)
- China(5)
- France(1)
- Germany(10)
- India(5)
- Italia (1)
- Japan(1)
- South Korea(1)
- Spain(2)
- Sweden(1)
- Switzerland(8)
- The Netherlands(2)
- United Kingdom(3)
- United States(86)
Contract Service Directory
Biozone Laboratories, Inc.
Company Headquarters
580 Garcia Ave.
Pittsburg, CA 94565
United States
Contract Service Directory
- Contract Manufacturing
- Formulation Development
- Lot Release Testing
- Manufacturing, Controlled Substances
- Manufacturing, Creams & Ointments
- Manufacturing, Custom
- Manufacturing, Gels
- Manufacturing, Generics
- Manufacturing, Liquids
- Manufacturing, OTC
- Manufacturing, Semisolids
- Manufacturing, Topicals
- Methods Development
- Methods Validation
- Microbiological Testing
- Microscopic Analysis
- Mixing & Blending
- Packaging, Creams & Ointments
- Packaging, Liquids
- Packaging, OTC
Related Content
-
Bio News | Biosimilars
Know the Challenges of Biosimilar Manufacturing
Best Practices for a Competitive MarketJerry Martin, Pharmaceutical and Life Sciences Consultant to PMMI, and, Dr. Michiel Ultee, Principal, Ulteemit BioConsulting, LLC 04.11.18
-
cGMP Manufacture | Drug Delivery | Industry News
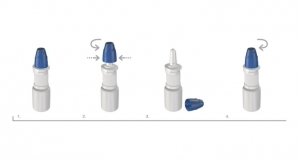
Aptar Pharma Extends CR/SF Mfg. Capabilities in North America
Installs manufacturing capacity for Child-Resistant and Senior-Friendly Classic Nasal Pump at Congers, NY site06.08.17
-
Breaking News | Drug Delivery | Industry News | Inspections
Tapemark Passes Regulatory Hurdle
Manufacturing facility completes successful FDA pre-approval and general GMP inspection06.07.17
-
Inspections | Regulatory Affairs
-
Breaking News | Industry News | Inspections

-
Bio News | Biologics, Proteins, Vaccines | Breaking News | cGMP Manufacture | Drug Development | Industry News

Lonza Acquires PharmaCell
Offers core expertise in autologous cell and gene therapy manufacturing06.05.17
-
Breaking News | Promotions & Moves

-
Breaking News | Facilities | Industry News | Inspections

-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Clinical Trial Materials | Clinical Trials | Industry News

Kemwell Provides Services for U.S. Trial
Supplies biologics drug substance for phase 1 clinical trials for U.S. IND filing05.18.17
-
Saneca Pharma Expands Services in Russia
Receives confirmation of multi-dosage cGMP approval for Russia05.16.17
-
Regulatory Affairs

The FDA and Metrics
The Agency and industry continue to work together to achieve FDA’s objectivesDavid Lonza, Lachman Consultant Services, Inc. 05.09.17
-
Breaking News | cGMP Manufacture | Industry News | Injectables | Parenterals | Solid Dosage/Creams/Ointments | Solid Dosage/Semi-solids

Fresenius Kabi Acquires Akorn in $4.3bn Deal
Strategically complementary combination to enhance and diversify Fresenius Kabi’s business and portfolio04.26.17
-
Trump Picks New FDA Commish
Combatting high drug prices and overregulation likely to be high on agendaTim Wright, Editor 04.03.17
-
Aseptic Processing | Fill/Finish | Injectables | Vials

Newsmakers: Peter Soelkner
A conversation with Peter Soelkner, managing director, Vetter Pharma International GmbHTim Wright, Contract Pharma 04.03.17
-
Capsules | Solid Dosage/Semi-solids
Generic Drug Trends
Industry faces tighter capacity, consolidation, and drug pricing uncertaintyEdward S. Price, President, PCI 03.07.17
-
Aseptic Processing | Bio News | Breaking News | Facilities | Fill/Finish | Industry News | Inspections | Lyophilization
LSNE Completes Two FDA Inspections
FDA PAI and general GMP inspection of two aseptic manufacturing plants and QC laboratory completed successfully03.06.17
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Drug Development | Industry News
Fujifilm Launches Bio CDMO Division
Aiming to achieve 100 billion yen in revenue by FY2024/March03.02.17
-
QA/QC | Regulatory Affairs

FDA’s Quality Agreements Guidance
Evaluation of FDA’s recently issued Contract Manufacturing Arrangements for Drugs: Quality Agreements Guidance for IndustryPaul Mason, Ph.D., Director of Lachman Consultant Services, Inc. 01.26.17











