Deborah Ensign, Microtest Laboratories01.22.13
Sterile pharmaceutical and medical device manufacturing environments require an effective cleaning and disinfection program to maintain aseptic conditions and prevent the microbial contamination of the product. The qualification of the chemical disinfectants used in these environments is extremely important, yet it is often overlooked. Disinfectant qualifications require more planning, time and resources than many companies realize. Considering the potential issues and difficulties that could occur while performing these qualifications, contracting an outside lab experienced in disinfectant qualifications may be the most efficient and least painful way to perform this work.
The following six steps provide a framework to assist companies in qualifying the disinfectants used in their environmental cleaning processes. Whether performed internally or by an outside testing lab, they must be addressed.
Step 1: Determine the Test Method
There are a number of methods for qualifying a disinfectant published by the Association of Official Analytical Chemists (AOAC), yet these are for qualifying the disinfectant itself. They are not appropriate for demonstrating the efficacy of a disinfectant within the pharmaceutical, biotechnology, and medical device industries.
Two of the most common methods suggested for disinfection qualification in these environments are:
Typically, standard American Type Culture Collection (ATCC) test organisms representing the basic classes of microorganisms (Gram negative, Gram positive, spore-former, fungus) along with actual environmental isolates from the client’s facility should be used in the qualification.

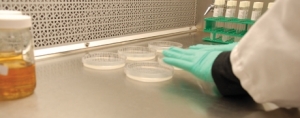
Examples of surface sample coupons (polypropylene,
vinyl, stainless steel, epoxy coated stainless steel)
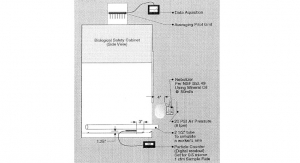
Step 3: Determine the Surface Types to be Tested
Each of the construction materials used in the clean room and/or other controlled areas should be tested separately. Examples of common materials are stainless steel, glass, plastic, and Plexiglas®. Normally 2-inch by 2-inch square coupons are used for the qualification.
All coupons must be sterilized or disinfected before use in the qualification. Depending on the material, sterilization may be accomplished through steam, ethylene oxide (EO), or chemical methods.
Step 4: Determine Expiration of Disinfectants
The qualification should replicate the same disinfectant concentration and contact exposure time used in the facility. It also should be performed using the worst-case expiration date for each disinfectant. If a container has a 30-day expiration once opened, and a dilution may be prepared and put into a spray bottle with an expiration of seven days, the efficacy testing should reflect this.

Examples of surface disinfectants
Step 5: Method Validation
Method validation is a critical step to verify that the testing method allows adequate recovery of the challenge organisms in the presence of the disinfectants. Regardless of the method being used, the test system must be inoculated with a low level of challenge organism, with and without (control) exposure to the disinfectant for the designated contact time.
Typically, the recovery of the challenge organisms should be within a factor of two of the positive controls for that organism. If the
recovery is not satisfactory, the testing method should be repeated using a different neutralization system and/or additional dilutions.
Step 6: Efficacy Testing
Efficacy testing is the actual testing of the disinfectant. Per the USP General Chapter <1072> Disinfectants, the test system is inoculated with sufficient inoculum to demonstrate at least a two log10 reduction for bacterial spores and a three log10 reduction for vegetative bacteria and allowed to dry. The inoculated system is then exposed to the desired concentration of the disinfectant for the desired contact time.

Swabbing the inoculated coupons
The surviving population in the test system is determined and the log10 reduction calculated. The log reduction data should be used to establish a scientifically supported disinfection program for the client’s facility.
Beyond the Six Steps
Any time a new disinfectant is introduced into the cleaning process within the facility, a qualification should be performed. From start to finish, a disinfectant qualification can require from two to 12 months to complete. The timeline will depend on a number of variables, including the number of disinfectants, contact time, and challenge organisms being tested as well as the number of surfaces (for the coupon method) being evaluated.
Deborah Ensign is supervisor, R&D Lab, at Microtest Laboratories, Inc. She has over 20 years of experience in regulatory microbiology and contract laboratory operations. For more information about this article, contact info@microtestlabs.com or 800-631-1680.
The following six steps provide a framework to assist companies in qualifying the disinfectants used in their environmental cleaning processes. Whether performed internally or by an outside testing lab, they must be addressed.
Step 1: Determine the Test Method
There are a number of methods for qualifying a disinfectant published by the Association of Official Analytical Chemists (AOAC), yet these are for qualifying the disinfectant itself. They are not appropriate for demonstrating the efficacy of a disinfectant within the pharmaceutical, biotechnology, and medical device industries.
Two of the most common methods suggested for disinfection qualification in these environments are:
- Tube method: This method evaluates disinfectants by inoculating dilutions of the disinfectant and determining the microbial reduction. It would most commonly be used as a simple screening to determine the type of disinfectant most effective against a specific set of organisms before performing a comprehensive disinfectant qualification.
- Coupon method: This method is more comprehensive and uses coupons made from actual facility surfaces. The surfaces are inoculated and exposed to the disinfectant. The inoculum is then removed from the surfaces and the log10 reduction determined.
Typically, standard American Type Culture Collection (ATCC) test organisms representing the basic classes of microorganisms (Gram negative, Gram positive, spore-former, fungus) along with actual environmental isolates from the client’s facility should be used in the qualification.
Examples of surface sample coupons (polypropylene,
vinyl, stainless steel, epoxy coated stainless steel)
Each of the construction materials used in the clean room and/or other controlled areas should be tested separately. Examples of common materials are stainless steel, glass, plastic, and Plexiglas®. Normally 2-inch by 2-inch square coupons are used for the qualification.
All coupons must be sterilized or disinfected before use in the qualification. Depending on the material, sterilization may be accomplished through steam, ethylene oxide (EO), or chemical methods.
Step 4: Determine Expiration of Disinfectants
The qualification should replicate the same disinfectant concentration and contact exposure time used in the facility. It also should be performed using the worst-case expiration date for each disinfectant. If a container has a 30-day expiration once opened, and a dilution may be prepared and put into a spray bottle with an expiration of seven days, the efficacy testing should reflect this.
Examples of surface disinfectants
Step 5: Method Validation
Method validation is a critical step to verify that the testing method allows adequate recovery of the challenge organisms in the presence of the disinfectants. Regardless of the method being used, the test system must be inoculated with a low level of challenge organism, with and without (control) exposure to the disinfectant for the designated contact time.
Typically, the recovery of the challenge organisms should be within a factor of two of the positive controls for that organism. If the
recovery is not satisfactory, the testing method should be repeated using a different neutralization system and/or additional dilutions.
Step 6: Efficacy Testing
Efficacy testing is the actual testing of the disinfectant. Per the USP General Chapter <1072> Disinfectants, the test system is inoculated with sufficient inoculum to demonstrate at least a two log10 reduction for bacterial spores and a three log10 reduction for vegetative bacteria and allowed to dry. The inoculated system is then exposed to the desired concentration of the disinfectant for the desired contact time.
Swabbing the inoculated coupons
The surviving population in the test system is determined and the log10 reduction calculated. The log reduction data should be used to establish a scientifically supported disinfection program for the client’s facility.
Beyond the Six Steps
Any time a new disinfectant is introduced into the cleaning process within the facility, a qualification should be performed. From start to finish, a disinfectant qualification can require from two to 12 months to complete. The timeline will depend on a number of variables, including the number of disinfectants, contact time, and challenge organisms being tested as well as the number of surfaces (for the coupon method) being evaluated.
Deborah Ensign is supervisor, R&D Lab, at Microtest Laboratories, Inc. She has over 20 years of experience in regulatory microbiology and contract laboratory operations. For more information about this article, contact info@microtestlabs.com or 800-631-1680.