Eric S. Langer, BioPlan Associates06.02.15
Outsourcing of biomanufacturing has been on the rise in recent years; one measure of success among contract manufacturing organizations (CMOs) is their ability to ensure their facilities keep up with demand. CMO constraints are not necessarily an unhealthy indicator, but must be balanced against clients’ need for access to flexible scheduling, and capacity. In fact, results from BioPlan Associates’ newly-released 12th Annual Report and Survey of Biopharmaceutical Manufacturing Capacity and Production1 show that CMOs are facing capacity constraints to a more significant degree than in recent years, all while operating at high levels of capacity. And while perceptions of future capacity constraints are more optimistic, CMOs clearly believe that, among a broad swath of improvements they will be implementing, improving their downstream purification technologies will be paramount to overcoming bottleneck challenges.
What the Data Show
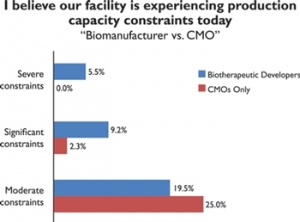
In our annual global study of 237 biopharmaceutical manufacturers and CMOs, a surprisingly high 36% of CMOs believe their facility is experiencing at least “significant” production capacity constraints today. And if we include those experiencing at least “moderate” constraints, roughly half of CMOs today are experiencing real capacity problems.
The data show some interesting trends when compared with last year’s results. Although last year about the same percentage of CMOs surveyed were experiencing at least “moderate” constraints, far more are finding constraints are worsening. This year almost twice as many report significant constraints (32.7% vs. 17.4% in 2014).
Unhealthy Capacity Utilization
Capacity constraints aren’t always a negative for CMOs, as many presume future growth is based on increasing demand. Further, when properly managed, constraints can be an indicator of efficiency, but must be balanced against clients’ need for access to flexible capacity.
On the other hand, this year’s survey results show CMOs may be hitting limits of what would be considered a healthy level of excess (flex) capacity. This is particularly the case for mammalian cell culture—the dominant paradigm—for which CMO respondents this year estimated currently operating at an average 82% of operating capacity. This is a hefty increase from last year’s average of 58% capacity utilization. The last time we saw levels in this range were 10 years ago during what was often referred to as an industry-wide capacity crunch.
Likewise, CMOs this year estimate operating at around 68% of microbial fermentation capacity, up from last year’s 54%. With CMOs operating at significantly higher levels of capacity this year than last, it’s easy to see why perceptions of capacity constraints are also trending up.
Future Capacity Constraints
Even so, it’s encouraging to see that CMO respondents to this year’s study are less likely to perceive significant capacity constraints in the medium-term than they are to be experiencing them today. Compared to the 36% experiencing significant or severe constraints today, only 1 in 5 are expecting that level of bottlenecks in 2020—essentially, CMOs are confident of their ability to manage capacity problems in the future.
Overall, our data indicates that about 8 in 10 CMOs expect to face some level of capacity constraint at their facility within 5 years’ time, down from roughly 9 in 10 last year. Along with that, fewer respondents this year expect severe or significant constraints in 5 years than did last year.
CMOs Spending on Expanding Facilities
Although CMOs this year are concerned about their current situation, they are more positive about their ability to handle future capacity constraint problems. This may be a reflection of anticipated spending on new facility construction: About 7 in 10 CMOs plan to increase their spending on new facilities this year, and the overall level of budget increases outpace expected spending hikes in many other areas, including staff hiring, process development, and new technologies for upstream and downstream production.
Moreover, CMOs this year estimate expanding their mammalian cell culture capacity by a substantial 77% over the next 5 years, double the overall amount of expansion (39%) they had forecast last year. Clearly, these organizations seeing that they are operating at high levels of mammalian production capacity today forecast expansions in the future that will ease their constraints.
CMOs may also be considering the current climate, in which, in contrast to the blockbuster approvals of the early 2000s, most of the current and upcoming drug approvals are for niche products with inherent smaller markets, additional indications of therapy on existing products, new countries, for sub-$1b markets, or for biosimilars/biogenerics production.
While the onset of biosimilars and biogenerics production is likely to favor CMOs, many of the new products in the pipeline can be absorbed into biomanufacturers’ existing facilities, as the manufacturing volumes are smaller. For biomanufacturers, whose expected future constraints are more moderate in nature than CMOs, there is less alarm, as there are now more options for retrofitting existing facilities to add incremental facility in a relatively short amount of time. And the option for outsourcing will of course continue to be attractive, too.
Avoiding Constraints
As part of our ongoing trend analysis of the causes—and potential alleviators—of capacity constraints, we asked CMOs to evaluate the factors likely to create production capacity constraints at their facility over the next 5 years.
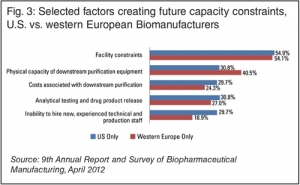
CMOs were most likely to report facility constraints (68%, up from 56% in 2014) as the source of capacity constraints, with this factor consistently atop the rankings over the past several years. In an interesting divergence from last year’s results, however, the physical capacity of downstream purification equipment was the factor second-most likely to be fingered as a culprit by CMOs. This factor was cited by a majority (53%) of respondents this year, up from 37% last year. That’s an important indicator, given improvements in purification technology that suppliers are investing in to move the industry away from such problems, including:
Beyond facility and downstream equipment constraints, CMOs also see staffing problems as contributing to future capacity constraints, with many pointing to an:
Unlike many other industries, biopharmaceutical manufacturing requires industry-specific experience around regulatory aspects, process variability, and high service level requirements. This makes it difficult to employ workers directly from related industries like chemical processing, food & beverage, etc. without significant additional (re)training.
Over the next 3-5 years, we expect to see experienced staff—especially those involved in operations and late-stage process development—become more sought after as the industry shifts to lower-cost manufacturing of biosimilars and other products in which flexibility and speed are especially important. Competition that will come about with the advent of biosimilars may cause biomanufacturers and CMOs to be less open about sharing best practices, leading them to place more importance on retaining key technical staff to at least prevent loss of knowledge to competitors.
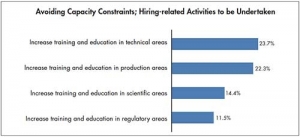
From our study, the inability to hire and retain experienced staff appears to be more of a concern for CMOs than for biomanufacturers. Not surprisingly, then, CMOs are also more likely to emphasize training and education as important areas to address in order to avoid significant capacity constraints. Compared to biomanufacturers, CMOs are more likely to see a pressing need to increase training and education in production, technical and regulatory areas, although there is less of a gap in the perceived need for training in scientific areas.
Even so, in the near-term, CMOs are most likely to push for the development of better downstream purification technologies to avoid future constraints. That’s to be expected, given that physical equipment constraints are a leading reason they expect those future bottlenecks. Single-use products are in high regard, with a significant portion of CMOs seeing the development of most cost-effective and/or better-performing single-use products as helping them avoid future constraints.
For the most part, CMOs and biomanufacturers are in agreement about the important areas to address in order to avoid future constraints, although in some instances there are some notable gaps between the two groups. According to our data, for example:
Conclusion
Our data indicates that a significant portion of CMOs, compared to biopharma companies, are experiencing capacity constraints today, perhaps as mammalian cell culture production capacity utilization rates trend higher. Although CMOs are increasing their new facility budgets, and data indicates that their mammalian cell culture capacity continues to grow year after year, one of the key culprits remains limitations with downstream purification, for which CMOs continue to seek improvements.
Newer and more efficient single-use products could help; this is in keeping with the general shift from blockbuster manufacture to manufacture of multiple niche products, but also is more generally suited to the CMO model, which involves juggling multiple products, platforms and processes. In fact, when we separately asked the industry to identify which new product development areas are of interest to them, the leading area for CMOs was disposable purification products, cited by almost half. Not far behind, chromatography products also rated highly, reflecting CMOs’ desire for more efficient downstream purification technologies.
Finally, while it’s easy to get caught up in talk of innovation, CMOs are also wary about staffing issues contributing to constraints in the medium-term. Training and educating key technical and operations staff will become more important to CMOs, as will retaining existing staff. After all, sophisticated process equipment needs sophisticated operators.
Eric S. Langer
BioPlan Associates
Eric S. Langer is president and managing partner at BioPlan Associates, Inc., a biotechnology and life sciences marketing research and publishing firm established in Rockville, MD in 1989. He is editor of numerous studies, including “Biopharmaceutical Technology in China,” “Advances in Large-scale Biopharmaceutical Manufacturing”, and many other industry reports. elanger@bioplanassociates.com 301-921-5979. www.bioplanassociates.com
What the Data Show
In our annual global study of 237 biopharmaceutical manufacturers and CMOs, a surprisingly high 36% of CMOs believe their facility is experiencing at least “significant” production capacity constraints today. And if we include those experiencing at least “moderate” constraints, roughly half of CMOs today are experiencing real capacity problems.
The data show some interesting trends when compared with last year’s results. Although last year about the same percentage of CMOs surveyed were experiencing at least “moderate” constraints, far more are finding constraints are worsening. This year almost twice as many report significant constraints (32.7% vs. 17.4% in 2014).
Unhealthy Capacity Utilization
Capacity constraints aren’t always a negative for CMOs, as many presume future growth is based on increasing demand. Further, when properly managed, constraints can be an indicator of efficiency, but must be balanced against clients’ need for access to flexible capacity.
On the other hand, this year’s survey results show CMOs may be hitting limits of what would be considered a healthy level of excess (flex) capacity. This is particularly the case for mammalian cell culture—the dominant paradigm—for which CMO respondents this year estimated currently operating at an average 82% of operating capacity. This is a hefty increase from last year’s average of 58% capacity utilization. The last time we saw levels in this range were 10 years ago during what was often referred to as an industry-wide capacity crunch.
Likewise, CMOs this year estimate operating at around 68% of microbial fermentation capacity, up from last year’s 54%. With CMOs operating at significantly higher levels of capacity this year than last, it’s easy to see why perceptions of capacity constraints are also trending up.
Future Capacity Constraints
Even so, it’s encouraging to see that CMO respondents to this year’s study are less likely to perceive significant capacity constraints in the medium-term than they are to be experiencing them today. Compared to the 36% experiencing significant or severe constraints today, only 1 in 5 are expecting that level of bottlenecks in 2020—essentially, CMOs are confident of their ability to manage capacity problems in the future.
Overall, our data indicates that about 8 in 10 CMOs expect to face some level of capacity constraint at their facility within 5 years’ time, down from roughly 9 in 10 last year. Along with that, fewer respondents this year expect severe or significant constraints in 5 years than did last year.
CMOs Spending on Expanding Facilities
Although CMOs this year are concerned about their current situation, they are more positive about their ability to handle future capacity constraint problems. This may be a reflection of anticipated spending on new facility construction: About 7 in 10 CMOs plan to increase their spending on new facilities this year, and the overall level of budget increases outpace expected spending hikes in many other areas, including staff hiring, process development, and new technologies for upstream and downstream production.
Moreover, CMOs this year estimate expanding their mammalian cell culture capacity by a substantial 77% over the next 5 years, double the overall amount of expansion (39%) they had forecast last year. Clearly, these organizations seeing that they are operating at high levels of mammalian production capacity today forecast expansions in the future that will ease their constraints.
CMOs may also be considering the current climate, in which, in contrast to the blockbuster approvals of the early 2000s, most of the current and upcoming drug approvals are for niche products with inherent smaller markets, additional indications of therapy on existing products, new countries, for sub-$1b markets, or for biosimilars/biogenerics production.
While the onset of biosimilars and biogenerics production is likely to favor CMOs, many of the new products in the pipeline can be absorbed into biomanufacturers’ existing facilities, as the manufacturing volumes are smaller. For biomanufacturers, whose expected future constraints are more moderate in nature than CMOs, there is less alarm, as there are now more options for retrofitting existing facilities to add incremental facility in a relatively short amount of time. And the option for outsourcing will of course continue to be attractive, too.
Avoiding Constraints
As part of our ongoing trend analysis of the causes—and potential alleviators—of capacity constraints, we asked CMOs to evaluate the factors likely to create production capacity constraints at their facility over the next 5 years.
CMOs were most likely to report facility constraints (68%, up from 56% in 2014) as the source of capacity constraints, with this factor consistently atop the rankings over the past several years. In an interesting divergence from last year’s results, however, the physical capacity of downstream purification equipment was the factor second-most likely to be fingered as a culprit by CMOs. This factor was cited by a majority (53%) of respondents this year, up from 37% last year. That’s an important indicator, given improvements in purification technology that suppliers are investing in to move the industry away from such problems, including:
- Moving bed technologies;
- Fast-flow resins;
- Greater binding capacity resins;
- Membrane separation technologies;
- Better UF-membranes; and
- More single-use equipment for purification.
Beyond facility and downstream equipment constraints, CMOs also see staffing problems as contributing to future capacity constraints, with many pointing to an:
- Inability to retain experienced technical and production staff (42%);
- Inability to hire new, experienced technical and production staff (37%); and an
- Inability to hire new, experienced scientific staff (32%).
Unlike many other industries, biopharmaceutical manufacturing requires industry-specific experience around regulatory aspects, process variability, and high service level requirements. This makes it difficult to employ workers directly from related industries like chemical processing, food & beverage, etc. without significant additional (re)training.
Over the next 3-5 years, we expect to see experienced staff—especially those involved in operations and late-stage process development—become more sought after as the industry shifts to lower-cost manufacturing of biosimilars and other products in which flexibility and speed are especially important. Competition that will come about with the advent of biosimilars may cause biomanufacturers and CMOs to be less open about sharing best practices, leading them to place more importance on retaining key technical staff to at least prevent loss of knowledge to competitors.
From our study, the inability to hire and retain experienced staff appears to be more of a concern for CMOs than for biomanufacturers. Not surprisingly, then, CMOs are also more likely to emphasize training and education as important areas to address in order to avoid significant capacity constraints. Compared to biomanufacturers, CMOs are more likely to see a pressing need to increase training and education in production, technical and regulatory areas, although there is less of a gap in the perceived need for training in scientific areas.
Even so, in the near-term, CMOs are most likely to push for the development of better downstream purification technologies to avoid future constraints. That’s to be expected, given that physical equipment constraints are a leading reason they expect those future bottlenecks. Single-use products are in high regard, with a significant portion of CMOs seeing the development of most cost-effective and/or better-performing single-use products as helping them avoid future constraints.
For the most part, CMOs and biomanufacturers are in agreement about the important areas to address in order to avoid future constraints, although in some instances there are some notable gaps between the two groups. According to our data, for example:
- CMOs are more likely to see the value in establishing manufacturing standards and industry benchmarks, as well as the aforementioned training and education efforts; while
- Biomanufacturers are more apt to feel the industry should develop more “modularized” production systems and fund more research to maximize production efficiencies.
Conclusion
Our data indicates that a significant portion of CMOs, compared to biopharma companies, are experiencing capacity constraints today, perhaps as mammalian cell culture production capacity utilization rates trend higher. Although CMOs are increasing their new facility budgets, and data indicates that their mammalian cell culture capacity continues to grow year after year, one of the key culprits remains limitations with downstream purification, for which CMOs continue to seek improvements.
Newer and more efficient single-use products could help; this is in keeping with the general shift from blockbuster manufacture to manufacture of multiple niche products, but also is more generally suited to the CMO model, which involves juggling multiple products, platforms and processes. In fact, when we separately asked the industry to identify which new product development areas are of interest to them, the leading area for CMOs was disposable purification products, cited by almost half. Not far behind, chromatography products also rated highly, reflecting CMOs’ desire for more efficient downstream purification technologies.
Finally, while it’s easy to get caught up in talk of innovation, CMOs are also wary about staffing issues contributing to constraints in the medium-term. Training and educating key technical and operations staff will become more important to CMOs, as will retaining existing staff. After all, sophisticated process equipment needs sophisticated operators.
Eric S. Langer
BioPlan Associates
Eric S. Langer is president and managing partner at BioPlan Associates, Inc., a biotechnology and life sciences marketing research and publishing firm established in Rockville, MD in 1989. He is editor of numerous studies, including “Biopharmaceutical Technology in China,” “Advances in Large-scale Biopharmaceutical Manufacturing”, and many other industry reports. elanger@bioplanassociates.com 301-921-5979. www.bioplanassociates.com