S. Harachand, Contributing Editor03.09.18
Big names in the global pharma packaging industry are expanding their base in India. Schott Kaisha announced the opening of its second production module at the company’s manufacturing facility in Jambusar, in the western Indian state of Gujarat in December last year. With this expansion, the German packaging major will up its current production capacity by almost 50% to more than three billion containers per year.
A leading supplier of glass syringes and cartridges for Indian drug makers, Schott Kaisha expects the new facility to run in full capacity by the end of this year.
With an objective to make India a leading export hub in manufacturing technology, Schott recently, launched new opto-electronic inspection protocol systems for vials. The advanced cosmetic inspection system covers 100% of the entire vial.
Close on the heels of Schott, Datwyler Sealing Solutions came out saying that the Swiss firm was preparing to commence production of pharmaceutical rubber components at a new plant in Satara, Maharashtra.
Datwyler presently manufactures around four billion rubber components used in vials, pre-filled syringes, and diagnostic devices at the Satara unit. The expansion of the project, which is scheduled to be completed by second quarter of 2018, would increase the production volume to five billion rubber components per annum by more than doubling the entire area of production.
The new facility would produce rubber vial and syringe components with fluoropolymer coating to prevent any interaction with the drugs, reports said.
India is emerging as one of the most important global manufacturing destinations, particularly in generics and biotech, Datwyler said while announcing the decision.
A rapidly expanding generic market, along with technological advancements and stringent regulations, is propelling the growth of the pharmaceutical packaging market the world over. The global pharma packaging market is expected to reach $104,882 million by 2022 from $68,749 million in 2015, growing at a compound annual growth rate (CAGR) of 6.3%, according to a report by Allied Market Research.
India’s pharma packaging market is growing at a faster pace, driven by the need for innovative packaging solutions to thrive in export markets, informal reports suggest.
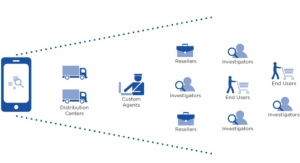
Systech International, a serialization company announced a strategic agreement in November last year, with Ahmedabad-based pharmaceutical packaging machinery maker NKP Pharma to provide software and hardware solutions to the Indian pharmaceutical industry that meet serialization and traceability regulations.
Systech offers the full stack of Level 1 to Level 5 solutions. Making use of NKP’s hardware and integration services, Systech can provide the Indian industry with complete and unified serialization solutions that meet audit and validation requirements, according to a news release.
Companies serving global markets have to ensure compliance as region-specific serialization regulations, like the Drug Supply Chain Security Act (DSCSA) in the U.S., require drug makers to comply with track and trace regulations to safeguard the pharma supply chain.
The EU has set the deadline of February 2019 for serialized authentication of pharma products. Many of the emerging markets including Russia, Australia, Taiwan, Egypt and Saudi Arabia are reportedly working on track and trace regulation.
India put in place procedures relating to tracking and tracing of export consignment of pharmaceuticals using bar code technology in 2014 by amending then existing provisions. India’s Directorate General of Foreign Trade (DGFT), under the ministry of commerce and industry, mandated that labelling covering the global trade item number (GTIN), batch number, expiry date and a unique serial number to be available on the primary packaging in human-readable form.
Primary packaging is the package that is in direct physical contact with the drug, secondary packaging is the carton containing multiple primary packs including a mono carton, and tertiary packaging means a shipper containing multiple secondary packs, according to the notification issued by the commerce ministry.
For secondary level packaging, manufacturers are required to print a one or two dimensional barcode encoding unique and universal global product identification code in the format of a 14 digit GTIN along with batch number, expiry date and a unique serial number of the secondary pack.
On the tertiary level, the ministry mandated incorporation of one dimensional barcode encoding GTIN along with batch number, expiry date and a unique serial number—Serial
Shipping Container Code (SSCC).
DGFT extended the deadline for compliance further to March 31, 2017. However, there are still several manufacturers, especially from the small-scale sector, that have yet to fully implement the track-and-trace bar-coding requirements. Nevertheless, the government decided not to take any punitive action against them.
S. Harachand
Contributing Editor
S. Harachand is a pharmaceutical journalist based in Mumbai. He can be reached at harachand@gmail.com.
A leading supplier of glass syringes and cartridges for Indian drug makers, Schott Kaisha expects the new facility to run in full capacity by the end of this year.
With an objective to make India a leading export hub in manufacturing technology, Schott recently, launched new opto-electronic inspection protocol systems for vials. The advanced cosmetic inspection system covers 100% of the entire vial.
Close on the heels of Schott, Datwyler Sealing Solutions came out saying that the Swiss firm was preparing to commence production of pharmaceutical rubber components at a new plant in Satara, Maharashtra.
Datwyler presently manufactures around four billion rubber components used in vials, pre-filled syringes, and diagnostic devices at the Satara unit. The expansion of the project, which is scheduled to be completed by second quarter of 2018, would increase the production volume to five billion rubber components per annum by more than doubling the entire area of production.
The new facility would produce rubber vial and syringe components with fluoropolymer coating to prevent any interaction with the drugs, reports said.
India is emerging as one of the most important global manufacturing destinations, particularly in generics and biotech, Datwyler said while announcing the decision.
A rapidly expanding generic market, along with technological advancements and stringent regulations, is propelling the growth of the pharmaceutical packaging market the world over. The global pharma packaging market is expected to reach $104,882 million by 2022 from $68,749 million in 2015, growing at a compound annual growth rate (CAGR) of 6.3%, according to a report by Allied Market Research.
India’s pharma packaging market is growing at a faster pace, driven by the need for innovative packaging solutions to thrive in export markets, informal reports suggest.
Systech International, a serialization company announced a strategic agreement in November last year, with Ahmedabad-based pharmaceutical packaging machinery maker NKP Pharma to provide software and hardware solutions to the Indian pharmaceutical industry that meet serialization and traceability regulations.
Systech offers the full stack of Level 1 to Level 5 solutions. Making use of NKP’s hardware and integration services, Systech can provide the Indian industry with complete and unified serialization solutions that meet audit and validation requirements, according to a news release.
Companies serving global markets have to ensure compliance as region-specific serialization regulations, like the Drug Supply Chain Security Act (DSCSA) in the U.S., require drug makers to comply with track and trace regulations to safeguard the pharma supply chain.
The EU has set the deadline of February 2019 for serialized authentication of pharma products. Many of the emerging markets including Russia, Australia, Taiwan, Egypt and Saudi Arabia are reportedly working on track and trace regulation.
India put in place procedures relating to tracking and tracing of export consignment of pharmaceuticals using bar code technology in 2014 by amending then existing provisions. India’s Directorate General of Foreign Trade (DGFT), under the ministry of commerce and industry, mandated that labelling covering the global trade item number (GTIN), batch number, expiry date and a unique serial number to be available on the primary packaging in human-readable form.
Primary packaging is the package that is in direct physical contact with the drug, secondary packaging is the carton containing multiple primary packs including a mono carton, and tertiary packaging means a shipper containing multiple secondary packs, according to the notification issued by the commerce ministry.
For secondary level packaging, manufacturers are required to print a one or two dimensional barcode encoding unique and universal global product identification code in the format of a 14 digit GTIN along with batch number, expiry date and a unique serial number of the secondary pack.
On the tertiary level, the ministry mandated incorporation of one dimensional barcode encoding GTIN along with batch number, expiry date and a unique serial number—Serial
Shipping Container Code (SSCC).
DGFT extended the deadline for compliance further to March 31, 2017. However, there are still several manufacturers, especially from the small-scale sector, that have yet to fully implement the track-and-trace bar-coding requirements. Nevertheless, the government decided not to take any punitive action against them.
S. Harachand
Contributing Editor
S. Harachand is a pharmaceutical journalist based in Mumbai. He can be reached at harachand@gmail.com.