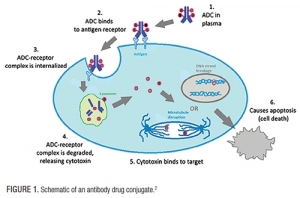
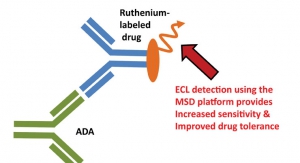
01.02.07
Daiichi Sankyo, Inc. has filed an sNDA with the FDA for WelChol to improve glycemic control in patients with type 2 diabetes mellitus. If approved, WelChol would be the first LDL cholesterol-lowering drug that also improves glycemic control.
WelChol has been shown to lower A1C in patients with type 2 diabetes mellitus. WelChol, when added to insulin, demonstrated a mean A1C reduction of 0.5% compared to placebo. In an additional WelChol study, the Glucose Lowering Effect Of WelChol Study (GLOWS), WelChol added to an oral antidiabetic therapy resulted in mean A1C reductions of 0.5% compared to placebo and 1% in patients with A1C = 8% compared to placebo.
The ADA estimates that 20.8 million people in the U.S. have diabetes, of which more than 90% have type 2 diabetes. The ADA recommends that patients with type 2 diabetes target an A1C level of less than 7%. Additionally, the National Cholesterol Education Program (NCEP) recommends that patients with type 2 diabetes keep their cholesterol levels in check and target an LDL-C goal of less than 100 mg/dL.
WelChol has been shown to lower A1C in patients with type 2 diabetes mellitus. WelChol, when added to insulin, demonstrated a mean A1C reduction of 0.5% compared to placebo. In an additional WelChol study, the Glucose Lowering Effect Of WelChol Study (GLOWS), WelChol added to an oral antidiabetic therapy resulted in mean A1C reductions of 0.5% compared to placebo and 1% in patients with A1C = 8% compared to placebo.
The ADA estimates that 20.8 million people in the U.S. have diabetes, of which more than 90% have type 2 diabetes. The ADA recommends that patients with type 2 diabetes target an A1C level of less than 7%. Additionally, the National Cholesterol Education Program (NCEP) recommends that patients with type 2 diabetes keep their cholesterol levels in check and target an LDL-C goal of less than 100 mg/dL.