YOU SEARCHED BY...
SEARCH BY...
Contract Service Directory
Country
- Argentina(1)
- Australia(8)
- Austria(1)
- Belgium(3)
- Canada(60)
- China(20)
- Denmark(2)
- Estonia(2)
- Finland(1)
- France(13)
- Germany(38)
- Greece(2)
- Hong Kong(1)
- Hungary(3)
- India(75)
- Ireland(1)
- Israel(3)
- Italia (6)
- Japan(2)
- Latvia(1)
- Lithuania(1)
- Mexico(2)
- New Zealand(1)
- Peru(1)
- Poland(2)
- Portugal(3)
- Romania(1)
- Singapore(1)
- Slovenia(2)
- South Korea(6)
- Spain(15)
- Sweden(3)
- Switzerland(22)
- Taiwan(3)
- The Czech Republic(1)
- The Netherlands(5)
- Turkey(2)
- Ukraine(1)
- United Kingdom(51)
- United States(674)
- Vietnam(1)
Contract Service Directory
Avior Risk and Compliance Assessment and Management
Company Headquarters
11 Perimeter Rd.
Nashua, NH 03063
United States
Contract Service Directory
Related Content
-
Promotions & Moves
Akebia Therapeutics Names CMO
Jain to lead clinical development of the company’s HIF pipeline05.25.17
-

Clean Air and Containment Decision Map
A risk assessment for the laboratory space should be prepared by, for example, the institution’s Biological Safety Officer.
-
Regulatory Affairs

Leveraging Next-Gen FSP Models to Better Manage Post-approval Regulatory Workloads
One-team Model forges an outsourcing partnership to deliver efficient global regulatory lifecycle managementElizabeth Madichie, Alistair Davidson, Denise Moody and Vincenzo Cannizzaro, PPD 05.09.17
-
Injectable Drug Delivery Trends
Graham Reynolds of West discusses the latest drug delivery strategies and trendsKristin Brooks, Contract Pharma 04.20.17
-
Analytical Services
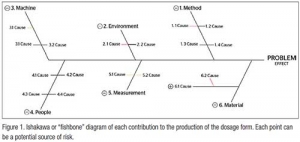
Quality Risk Management
It’s not just for the big guys anymoreEmil W. Ciurczak, DoraMaxx Consulting 04.03.17
-
Elemental Impurities: A Virtual Company Perspective
A look at risk assessments for elemental impurity determinations in oral tablet and parenteral drug products.Anthony DeStefano and Thomas Kester, Recordati Rare Diseases, Inc. 04.03.17
-
Clinical Trials | CRO News | Drug Development
ICON, Pfizer Ink Services Pact
To support Pfizer’s global drug development efforts for three years06.10.16
-
-
Promotions & Moves
Avion Pharma Names CEO
Current chief operating officer Harold Deas to assume chief exec role04.12.16
-
Breaking News | Clinical Trial Materials | Clinical Trials | Collaborations & Alliances | Preclinical Outsourcing
Hemispherx Enters Agreement With myTomorrows
Will focus on early access program for rintatolimod in Europe08.10.15
-











