YOU SEARCHED BY...
- Contract Services Directory
- Automated Pharmacology
- Accelerated Stability Studies
- Informatics
- Packaging, Liquids
- Packaging, Blister
- Manufacturing, Creams & Ointments
- IT, LIMS
- Chemistry, Combinatorial
- Chemical Analysis
- Packaging, Clinical Trials
- Process R&D
- Sustained Release Delivery Systems
- Contract Development and Manufacturing Organization (CDMO)
- Manufacturing, Creams & Ointments
- Manufacturing, Clinical Trials Supply
- Manufacturing, API, High Potency
SEARCH BY...
Country
Contract Service Directory
Meridian Life Science, Inc.
Company Headquarters
5171 Wilfong Road
Memphis, TN 38134
United States
Contract Service Directory
- Analytical Methods Development
- Analytical Validation Studies
- Aseptic Processing
- Assay Development
- Biodiagnostic Process Development
- Biologics Manufacturing
- Biologics Process Development
- Bioprocess Validation
- Bioprocessing
- Bioprocessing, Disposable/Single-Use
- Cell Culture Media Development
- Cell Line Construction & Development
- CMC (Chemistry, Manfacturing & Controls)
- Contract Development and Manufacturing Organization (CDMO)
- Contract Manufacturing
- Contract Research
- Custom Manufacturing
- Downstream Processing
- Endotoxin Testing & Removal
- Filling, Vial
- Filtration Services
- Fluid Bed Processing
- Formulation Development
- Formulation Services
- Gene Therapy & Testing Protocols
- Immunobiology Services
- Immunotoxicity Testing
- Manufacturing, Aseptic Fill/Finish
- Manufacturing, Clinical Trials Supply
- Manufacturing, Injectables
- Manufacturing, Proteins
- Manufacturing, Vaccines
- Methods Development
- Methods Validation
- Mycoplasma Testing
- Parenterals, Vial Filling
- Pilot-Scale Filling
- Process Development/Scale-Up Services
- Process R&D
- Process Validation Studies
- Protein Extraction/Purification
- Purification
- QA/QC
- R&D Services
- Scale-Up
- Stability Studies
- Sterile Filling
- Sterility Testing
- Technology Transfer
- Virology
Related Content
-
Promotions & Moves

CROS NT Appoints President of North American Ops
Paul has more than 25 years of experience serving customers and managing clinical trials05.30.17
-
APIs | cGMP Manufacture | Industry News

Cambrex Expands Capacity and Capabilites in Sweden
Installs new multi-purpose reactors, adds continuous flow technology at Karlskoga Facility05.30.17
-
cGMP Manufacture | Clinical Trial Materials | Formulation Development | Industry News
Quotient Clinical Appoints Chief Commercial Officer
Kalim to support further expansion of global commercial footprint05.30.17
-
Breaking News | Promotions & Moves

-
Breaking News | Drug Development | Drug Discovery | Facilities | Industry News

-
Biologics, Proteins, Vaccines | cGMP Manufacture | Drug Development | Industry News
Hitachi Chemical Acquires Remaining Stake in PCT
Expands services in the U.S. and adds services in Japan, Europe and other regions05.19.17
-

-
Cold Chain Management | Industry News | Logistics
Cold Chain Technologies Opens New HQ
Larger facility houses a manufacturing operation supported by thermal package engineering and an R&D center04.19.17
-
Biologics, Proteins, Vaccines | cGMP Manufacture | Industry News

-
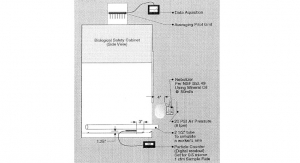
The Bio-Analog Test For Field Validation of a Biosafety Cabinet Performance
Acumen White Papers - Volume 4 No. 1
-
Breaking News | Drug Discovery | Industry News | Methods Development











