05.14.09
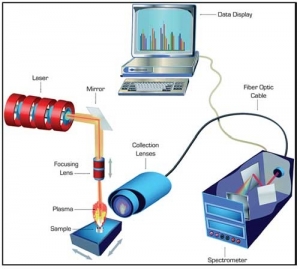
Microtest Laboratories is adding dissolution testing to its current suite of analytical testing and stability testing services. Dissolution is a compendial method used to measure a drug’s rate of release from its dosage form. It is a complement to other analytical assays — potency and related substances — that characterize the dosage form.
“The addition of dissolution testing is the perfect complement to our portfolio of services,” said Steven Richter, Ph.D., president and scientific founder of Microtest Laboratories. “By offering an even more comprehensive array of assays, we’re increasing the convenience, efficiency, and value that we deliver as a single, preferred provider of contract laboratory services.”
Dissolution is typically used in a number of settings, including: optimization of drug delivery rate during development studies; routine QC analysis for lot release and measurement of lot-to-lot variability; stability studies to examine the effects of time, temperature, and humidity on the dosage form; comparison of one dosage form to another; and estimated release and absorption rates in humans after an in vitro - in vivo correlation is established.
Dissolution is a sample preparation technique. Samples still need to be analyzed separately. Typical samples are oral dosage forms, although it can also be used on topicals and transdermal systems. Microtest’s analytical testing processes follow guidelines from the United States Pharmacopeia (USP), the Japanese and European Pharmacopoeias, the FDA, the International Organization for Standardization (ISO), and the Association for Advancement of Medical Instrumentation (AAMI).
“The addition of dissolution testing is the perfect complement to our portfolio of services,” said Steven Richter, Ph.D., president and scientific founder of Microtest Laboratories. “By offering an even more comprehensive array of assays, we’re increasing the convenience, efficiency, and value that we deliver as a single, preferred provider of contract laboratory services.”
Dissolution is typically used in a number of settings, including: optimization of drug delivery rate during development studies; routine QC analysis for lot release and measurement of lot-to-lot variability; stability studies to examine the effects of time, temperature, and humidity on the dosage form; comparison of one dosage form to another; and estimated release and absorption rates in humans after an in vitro - in vivo correlation is established.
Dissolution is a sample preparation technique. Samples still need to be analyzed separately. Typical samples are oral dosage forms, although it can also be used on topicals and transdermal systems. Microtest’s analytical testing processes follow guidelines from the United States Pharmacopeia (USP), the Japanese and European Pharmacopoeias, the FDA, the International Organization for Standardization (ISO), and the Association for Advancement of Medical Instrumentation (AAMI).