YOU SEARCHED BY...
SEARCH BY...
Contract Service Directory
Country
- Argentina(1)
- Australia(8)
- Austria(1)
- Belgium(3)
- Canada(59)
- China(20)
- Denmark(2)
- Estonia(2)
- Finland(1)
- France(13)
- Germany(40)
- Greece(2)
- Hong Kong(1)
- Hungary(3)
- India(76)
- Ireland(1)
- Israel(3)
- Italia (6)
- Japan(2)
- Latvia(1)
- Lithuania(1)
- Mexico(2)
- New Zealand(1)
- Peru(1)
- Poland(2)
- Portugal(3)
- Romania(1)
- Singapore(1)
- Slovenia(2)
- South Korea(6)
- Spain(15)
- Sweden(3)
- Switzerland(22)
- Taiwan(3)
- The Czech Republic(1)
- The Netherlands(5)
- Turkey(2)
- Ukraine(1)
- United Kingdom(51)
- United States(677)
- Vietnam(1)
Contract Service Directory
Norwich Pharma Services
Company Headquarters
6826 State Highway 12
Norwich, NY 13815
United States
Contract Service Directory
- Coating & Lamination
- Contract Development and Manufacturing Organization (CDMO)
- Contract Manufacturing
- Contract Packaging
- Encapsulation
- Fluid Bed Processing
- Granulation
- Manufacturing, Capsules
- Manufacturing, Controlled Substances
- Manufacturing, Oral Drug Delivery
- Manufacturing, Solid Dosage
- Manufacturing, Sustained Release
- Manufacturing, Tablets
- Packaging, Solid Dosage
- Tablet Coating
Content From Norwich Pharma Services
-
DEA Imposes Extra Layer of Regulations
on Manufacturers of FDA-Approved Prescription Drugs To Alleviate PainWhite Papers / Tech Papers Released on 07.17.2014
-
Solvent Considerations in Solid-Dose Manufacturing
...White Papers / Tech Papers Released on 02.06.2014
-
The Global Pharmacovigilance Landscape—Considerations Beyond the US and the EU
...White Papers / Tech Papers Released on 02.04.2014
-
Norwich Pharma Services Rebrands
Terry Novak of Norwich Pharma Services talks about the CDMO's rebranding.Videos Released on 11.13.2013
-
Norwich Pharmaceuticals' Business Surges In Second Half of 2013
...News Releases Released on 10.16.2013
-
The Global Pharmacovigilance Landscape—Considerations Beyond the US and the EU
...White Papers / Tech Papers Released on 10.25.2012
-
Solvent Considerations in Solid-Dose Manufacturing
...White Papers / Tech Papers Released on 06.20.2012
Related Content
-
-
APIs | Bio News | Breaking News | cGMP Manufacture | Collaborations & Alliances | Industry News | Supply Chain
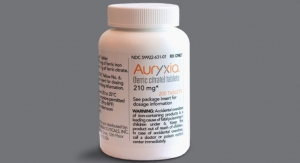
BioVectra, Keryx Enter Supply Agreement for Kidney Disease
The partnership revolves around the manufacture of Ferric Citrate, the API in Auryxia tablets12.14.17
-
Collaborations & Alliances | Packaging & Tracking | Serialization

Systech, NKP Pharma Partner to Expand Presence in India
The goal of the collaboration is unified serialization solutions in India that also meet various requirements11.22.17
-
Breaking News | Industry News

Quotient Sciences Acquires CDMO Pharmaterials
The move further expands Quotient’s footprint in the U.K.11.14.17
-
Industry News | Information Technology | Reader Showcase

Lonza Releases Latest MODA Software
Updated to allow companies to meet changing QC regulatory guidance10.25.17
-
Drug Delivery Innovation
Growth in biologics is driving advanced drug delivery technology solutionsTim Wright, Editor 10.11.17
-
Promotions & Moves

Envigo Appoints New Business Lead Consultant
Rehman will provide specialist expertise to the team of regulatory consultants servicing the chemicals industry10.11.17
-
Promotions & Moves

Envigo Makes Executive Appointments
Boyd to serve as chief commercial officer, Muller to head EMEA operations09.21.17
-

-
APIs | Breaking News | Clinical Trial Materials | Facilities | Formulation Development | Industry News | Solid Dosage/Creams/Ointments

-
Drug Development | Industry News

Alcami, UNCW Partner on Courses for Pharmaceutical Sciences
Aims to provide students with an overview of the drug development process from concept to commercialization07.28.17











