12.12.17
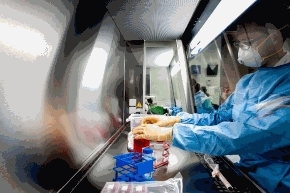
SGS has introduced a Sanger sequencing service at its Glasgow, UK, lab to support genetic stability testing and perform identity testing on cell banks, plasmids, and viral seeds/vectors. The new service, available under GMP conditions, is supported by an investment in ABI Life Technologies’ validated 3500xl DNA sequencing technology.
Sanger sequencing is a method of DNA sequencing based on the selective incorporation of chain-terminating deoxynucleotides by DNA polymerase during in vitro DNA replication. The technology has a range of applications including cell bank characterization, virus seed and plasmid identity testing and genetic stability testing of cell banks, and expands analysis and characterization solutions for the production and manufacturing of biopharmaceuticals.
“Biopharmaceutical manufacturing is evolving and growing at an incredible pace, but the focus of both clients and regulators is on the assurance of quality and the safety of patients,” said Dr. Archie Lovatt, scientific director at SGS Life Sciences. “By investing in the sequencing equipment and integrating this new service into our laboratories, we are expanding and complementing our capabilities, ensuring that SGS can offer its partners the widest range of skills, services, expertise and experience.”
The Glasgow facility was incorporated in 2012 following the acquisition of Vitrology Ltd., a biopharmaceutical contract testing organization specializing in biosafety testing. The facility was expanded in 2014 with an additional 500 m² of lab space.
Sanger sequencing is a method of DNA sequencing based on the selective incorporation of chain-terminating deoxynucleotides by DNA polymerase during in vitro DNA replication. The technology has a range of applications including cell bank characterization, virus seed and plasmid identity testing and genetic stability testing of cell banks, and expands analysis and characterization solutions for the production and manufacturing of biopharmaceuticals.
“Biopharmaceutical manufacturing is evolving and growing at an incredible pace, but the focus of both clients and regulators is on the assurance of quality and the safety of patients,” said Dr. Archie Lovatt, scientific director at SGS Life Sciences. “By investing in the sequencing equipment and integrating this new service into our laboratories, we are expanding and complementing our capabilities, ensuring that SGS can offer its partners the widest range of skills, services, expertise and experience.”
The Glasgow facility was incorporated in 2012 following the acquisition of Vitrology Ltd., a biopharmaceutical contract testing organization specializing in biosafety testing. The facility was expanded in 2014 with an additional 500 m² of lab space.