02.16.18
Cryoport has been selected by DiscGenics to provide logistics support to its multicenter, first-in-human study to evaluate the safety and preliminary efficacy of IDCT, its allogeneic injectable cell therapy, in patients with single level, symptomatic lumbar intervertebral disc degeneration.
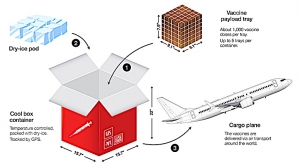
Cryoport's temperature-controlled logistics support services will include outbound logistics for DiscGenics' clinical phase 1/2 trial of IDCT to clinics in both the U.S., and eventually Japan.
"Degenerative disc disease can cause immense suffering for patients, yet current treatment options are limited," said Flagg Flanagan, chief executive officer, DiscGenics. "We are excited to commence clinical evaluation of IDCT, which utilizes proprietary therapeutic progenitor cells derived from adult human disc tissue to offer a non-surgical, potentially regenerative and cost-effective solution for the treatment of patients with mild to moderate degenerative disc disease. Using Cryoport's premium cold chain logistics solutions not only ensures we maintain superior quality assurance of our clinical trial materials when shipping both domestically and internationally, but also minimizes the risk of costly delays and clinical product loss during shipping as we progress through the trial."
Jerrell Shelton, chief executive officer, Cryoport, said, "DiscGenics is engaged in conducting clinical trials for their groundbreaking regenerative therapy for patients with chronic low back pain, and we are proud to be selected to ship their biological product and injection kits to clinical sites in the U.S. and Japan. Cryoport's reputation for reliability and safety were critical for DiscGenics when deciding to entrust its valuable cell therapies to a logistics partner. Our agreement is further evidence that Cryoport's chain of condition and chain of custody monitoring systems are the 'gold standard' in the life sciences industry with a wide variety of applications across the cellular therapy market."
Cryoport's temperature-controlled logistics support services will include outbound logistics for DiscGenics' clinical phase 1/2 trial of IDCT to clinics in both the U.S., and eventually Japan.
"Degenerative disc disease can cause immense suffering for patients, yet current treatment options are limited," said Flagg Flanagan, chief executive officer, DiscGenics. "We are excited to commence clinical evaluation of IDCT, which utilizes proprietary therapeutic progenitor cells derived from adult human disc tissue to offer a non-surgical, potentially regenerative and cost-effective solution for the treatment of patients with mild to moderate degenerative disc disease. Using Cryoport's premium cold chain logistics solutions not only ensures we maintain superior quality assurance of our clinical trial materials when shipping both domestically and internationally, but also minimizes the risk of costly delays and clinical product loss during shipping as we progress through the trial."
Jerrell Shelton, chief executive officer, Cryoport, said, "DiscGenics is engaged in conducting clinical trials for their groundbreaking regenerative therapy for patients with chronic low back pain, and we are proud to be selected to ship their biological product and injection kits to clinical sites in the U.S. and Japan. Cryoport's reputation for reliability and safety were critical for DiscGenics when deciding to entrust its valuable cell therapies to a logistics partner. Our agreement is further evidence that Cryoport's chain of condition and chain of custody monitoring systems are the 'gold standard' in the life sciences industry with a wide variety of applications across the cellular therapy market."