Search Results for 'Training'
-
Regulatory Affairs | Serialization

The DSCSA Delay What Does it Mean for the Pharma Industry?
What should companies be doing to implement a suitable serialization solution before the new enforcement date arrives?Erik Haeffler, Recipharm 09.06.17
-
Information Technology | Regulatory Affairs

Data Integrity: A Practical and Risk-Based Approach
Ways an organization can form a culture and cost-effective means to meet the various challenges in sustaining data integrity.Robert Marohn, ClinLogic, LLC 09.06.17
-
Regulatory Affairs | Serialization

The Psychology of DSCSA: Four Stages of Serialization
It’s time for manufacturers to adopt post-serialization efficiencies.Larry Hotz, Marketing Manager, WDPrx - Woodfield Pharmaceutical LLC 09.06.17
-
Cold Chain Management
Cold Chain Market and Technology Trends
Advanced technologies for cold chain management to ensure drug safety and efficacy continue to grow in importance.Mark Sawicki, Cryoport 09.06.17
-
Breaking News | Drug Development | Facilities | Formulation Development | Industry News

Colorcon Opens New Technology Services Laboratory in Vietnam
Marks the sixth such facility for Colorcon within Southeast Asia09.05.17
-
Drug Discovery

How Can We Predict Immunogenicity Earlier in Drug Discovery?
Identifying potential immune responses earlier in the development of biotherapeutics.Kevin Merlo and Tim Moran, Dassault Systèmes Biovia 09.01.17
-
Clinical Trials | Drug Development | Information Technology

Evolution of the CRO Project Manager
As clinical project managers become more strategic, more advanced technology must support their evolving rolesAndy Mehrotra, CEO, EightSpokes 08.31.17
-
Collaborations & Alliances | Drug Development | Process Development

MilliporeSigma, Angiex Enter Clinical Readiness Pact
MilliporeSigma to provide access to process development tools, education programs and training08.29.17
-
Biologics, Proteins, Vaccines | Collaborations & Alliances | Drug Development

MilliporeSigma Forms Strategic Vax Development Alliance
Aims to advance vaccine research and development for neglected and emerging infections08.08.17
-
Drug Development | Industry News

NSF International Adds Pharma Biotech Services in India
To offer pharma biotech consulting and training services from its Gurugram, India office08.08.17
-
Drug Development | Industry News

Alcami, UNCW Partner on Courses for Pharmaceutical Sciences
Aims to provide students with an overview of the drug development process from concept to commercialization07.28.17
-
Clinical Trials | Drug Development

CRO Competition Heats Up: Differentiate Offerings to Retain Site Loyalty
Site centricity becomes even more critical with increased CRO competition and M&ADave Espenshade, Vice President of CRO Partnerships, Greenphire 07.25.17
-
Drug Development | QA/QC | Regulatory Affairs

India Frames Guidance for Drug Surveillance
Outsourcing PV functions to riseS. Harachand, Contributing Editor 07.18.17
-
Regulatory Affairs

Data Integrity Guidance Around the World
A look at data integrity controls and the overall framework for a data governance system.James Davidson, Ph.D, Lachman Consultants 07.18.17
-
Promotions & Moves

-
Industry News | Serialization

Adents Implements Multi-Site, Multi-line Serialization Solution
Installs serialization solution on packaging lines at four sites in France06.08.17
-
Bio News | Biologics, Proteins, Vaccines | cGMP Manufacture | Industry News

NIBRT, GE Healthcare Open SU Bio-Mfg. CoE
Aims to drive next-generation biomanufacturing expertise with a new training center06.08.17
-
Biologics, Proteins, Vaccines

Trends in New Technologies & Budgets
Budget increases in biopharmaceutical industry indicate future growthEric S. Langer, BioPlan Associates 06.06.17
-
Regulatory Affairs | Serialization

Lessons Learned From Global Serialization
A look at some key findings leading up to Europe’s 2019 FMDCarlos Machado, Serialization Director, SEA Vision U.S. 06.06.17
-
Drug Development

Solving the Pharma Talent Crunch
Addressing talent shortages in today's pharmaceutical sectorJim Sykes, Managing Director, Pharmaceutical & Life Sciences, Alexander Mann Solutions 05.30.17
-

Transforming Sharp into a LEAN organization
Becoming a LEAN organisation is now at the forefront of every CMO’s agenda.
-
Clinical Trials | Drug Development | Information Technology | Regulatory Affairs
Using Pragmatic Randomization in Real-World Studies
How PCTs increase the robustness of real world studies at a fraction of the cost of classic randomized trialsNancy Dreyer, MPH, Ph.D. , Global Chief of Scientific Affairs and Senior Vice President, Real-World & Late Phase Research, QuintilesIMS 05.22.17
-

Sharp Packaging US Surpasses 50 Quality Certifications
Greg Ness, Quality Director for Sharp Packaging Solutions
-
Clinical Trials | Drug Development | Information Technology

Breaking New Ground: How Industry Collaboration is Transforming Clinical Research
An increasingly complex clinical trial landscape drives the life sciences industry to support broad collaborationsJill Johnston, Vice President, Veeva Vault, Veeva Systems 05.15.17
-
Supply Chain

Strengthen Your Supply Chain
S&OP is critical to optimal decision making in today’s changing pharmaceutical industryDavid Medina, Vice President - Solutions and Vertical Markets, QAD, Inc. 05.09.17
-
Clinical Trials

Tracking Health
Exploring the impact of wearable health monitoring tools on CROsNigel Walker 05.09.17
-
Analytical Services | Bioanalytical Services | R&D

CROs & Next-Gen Drug Development
R&D trends drive outsourcing in today’s marketKristin Brooks, Contract Pharma 05.09.17
-
Bio News | cGMP Manufacture | Clinical Trial Materials | Industry News | Scale-up/Technology Transfer

United BioPharma Selects GE’s FlexFactory
To build late-stage clinical and commercial production capacity for biopharmaceuticals04.25.17
-
Bio News | Biologics, Proteins, Vaccines | Drug Development | Industry News | Laboratory Testing

MilliporeSigma, LabCentral to Foster Biotech Startups
MilliporeSigma to provide access to process development tools, education programs and training04.25.17
-
Biologics, Proteins, Vaccines
Biomanufacturing Trends and Outlook 2017: Where Is It All Going?
A look at the technological advances permeating (bio)pharma manufacturingBen Locwin, Contributing Editor 04.20.17
-
Drug Delivery

Inventing the MDI: A History in Modern Inhalation Therapy
3M’s Charlie Thiel offers insight into the pressurized metered-dose inhalerOnline Exclusives Lynn Melling, Contributing Author 04.19.17
-
Biosimilars
Biosimilars Update
Fulfilling a dire need, but at what (human) cost?Ben Locwin, Contributing Editor 04.03.17
-
Elemental Impurities: A Virtual Company Perspective
A look at risk assessments for elemental impurity determinations in oral tablet and parenteral drug products.Anthony DeStefano and Thomas Kester, Recordati Rare Diseases, Inc. 04.03.17
-
Industry News | Information Technology
Enterprise System Partners & Werum IT Renew PAS-X Pact
Aims to ensure ESP employees are certified in the delivery of Werum’s PAS-X MES to customers in Europe and USA03.15.17
-
Bio News | Breaking News | Industry News
Cook Pharmica Teams Up With Ivy Tech Bloomington
Partnership to further rural biotechnology education03.08.17
-
Regulatory Affairs
Protecting Pharma Intellectual Property
SmartFile CEO offers ways to protect against theftJohn Hurley, SmartFile 03.07.17
-
Biologics, Proteins, Vaccines
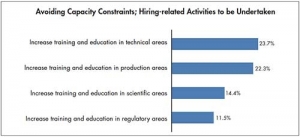
CMOs Hiring Constraints: Cell Therapy Manufacturing Expecting Future Challenges
Technically trained staff needed to run complex manufacturing platforms, automationEric S. Langer, BioPlan Associates 03.07.17
-
Regulatory Affairs | Serialization

Successful Serialization Partner Selection
Can speed, business value and total cost of ownership exist in the same space when compliance cannot wait?Michael Howe, CEO, Verify Brand 03.07.17
-
Regulatory Affairs | Serialization

DSCSA Serialization: It’s Not Too Late
If you’re not yet compliant with DSCSA, here are some practical steps to followJohn Jordon and Vito Pirrera, Vantage Consulting Group 03.07.17
-
Clinical Trials | Industry News | Information Technology
SGS Launches Simulo Clinical Trial Platform
Designed to facilitate complex clinical trial simulation to predict expected results02.24.17
-
Breaking News | Collaborations & Alliances | Industry News
Sartorius Partners With EMBL
Sartorius is now a member of the group of corporate EMBL partners aiming at connecting industry to academia02.21.17
-
Promotions & Moves
-
Analytical Services | Breaking News | Collaborations & Alliances | CRO News | Drug Discovery | Industry News
-
Aseptic Processing | cGMP Manufacture
-
Clinical Trials | Process Development
MilliporeSigma to Provide Development and Manufacturing Services to Y-mAbs
Includes scale-up and GMP manufacturing of drugs in late-stage development08.01.16
-
Promotions & Moves | Regulatory Affairs
Novella Clinical Adds Three Senior-level Hires
Markwood, Bala, Yucaitis to serve in oncology roles and regulatory affairs07.22.16
-
Clinical Trials | Information Technology | R&D
An Adoption Divide: CDISC Standards
Survey addresses challenges, advantages, and costs associated with implementing clinical data standardsDavid A. Evans, managing director quality governance and regulatory compliance, Accenture Life Sciences 07.20.16
-
Drug Development | Drug Discovery | Industry News | Laboratory Testing
MilliporeSigma to Build Life Science Campus in Burlington, MA
Will serve as a hub for Merck KGaA’s North American life science business07.20.16
-
Breaking News | Facilities | Industry News | Process Development
MilliporeSigma Relaunches Customer Collaboration Centers
Includes new concept and M Lab name07.18.16
-
Validation












