Search Results for 'contract pharma'
Contract Service Directory Companies
Solution Content Center Companies
View All Buyers Guide Companies »
-

Videobite: Contract Pharma Talks with Tony Kelleher of Actylis
Facility Director of Actylis Limerick discusses ensuring quality of biopharma raw materials and success stories adapting manufacturing processes.Videos
-

Videobite: Contract Pharma Talks with Tom Wilson of Pfizer CentreOne
Vice President and Global Contract Manufacturing Leader discusses capacity planning, pricing, and handling supply chain challenges.Videos
-

Videobite: Contract Pharma Sits Down with Jason Apter of Sannova Analytical
Sannova's CEO discusses the role CROs play in the drug development process, the role of AI, and strategies to accelerate drug development.Videos
-

Videobite: Contract Pharma Sits Down with Josselyn Haas Durr of MilliporeSigma
Manufacturing Science and Technology Manager discusses M Lab Collaboration Centers and addressing customers pain points.Videos
-

Videobite: Contract Pharma Sits Down with Bob Voelkner of LabVantage
Vice President of Sales & Marketing discusses data integrity, compliance, and best practices when evaluating and investing in digital transformation.Videos
-
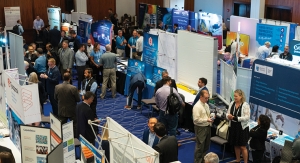
The 22nd Annual Contract Pharma Contracting & Outsourcing Conference and Tabletop exhibition
Contracting & Outsourcing 2023 featured a sold-out exhibit hall and informative conference sessions that were well attended.11.14.23
-

Videobite: Contract Pharma Sits Down with Alex Mitchell of Sentry BioPharma Services
Vice President of Operations discusses the role of a Qualified Person (QP) in the clinical pharmaceutical space.Videos
-

Videobite: Contract Pharma Sits Down with Alex Mitchell of Sentry BioPharma Services
Vice President of Operations discusses Foreign Trade Zone and how a pharmaceutical company can benefit from using an FTZ.Videos
-
cGMP Manufacture | Fill/Finish

Videobite: Contract Pharma Sits Down with Chris Preti of Jubilant HollisterStier
CEO and President discusses supporting pharma & biotech companies looking for complex fill/finish solutions.Videos
-
-
Aseptic Processing | Fill/Finish | Parenterals

The Contract Pharma Podcast: Val Pannell, Business Development Manager, GRAM
Grand River Aseptic Manufacturing's business development manager Val Pannell talks about the keys to successful CDMO and sponsor partnerships.09.01.23
-
Biologics, Proteins, Vaccines | cGMP Manufacture | Drug Development | VideoBites

Videobite: Contract Pharma Sits Down with Christoph Buerki of Novartis GlobalBiotech Cooperations
Head of Finance Operations & Contract Manufacturing discusses the company's strategic end to end CDMO services and manufacturing technologies.Videos
-
cGMP Manufacture | Fill/Finish | VideoBites

Videobite: Contract Pharma Sits Down with Val Pannell of GRAM
How CDMOs can address customers key concerns when partnering on projects.Videos
-
Clinical Trials | Drug Development | Logistics | Supply Chain

Reducing the Risk of Comparator Sourcing
Transitioning to a strategic sourcing approach.Contract Pharma 05.26.23
-
VideoBites

Videobite: Contract Pharma Sits Down with Ryan Lake of Societal CDMO
Discusses sterile injectable and OSD capabilities, recent FDA approval to manufacture commercial tablet, and Pharma supply chain trends and challenges.Videos
-
Breaking News

Contract Pharmaceuticals Limited Appoints Jan Sahai as CEO
Sahai joined CPL in 2005 as head of business development.01.20.23
-
cGMP Manufacture | Drug Delivery | Drug Development

Contract Pharma Q&A with Jeffrey Speicher of Avéma Pharma Solutions
Provides insight on industry trends, outsourcing, and growth opportunities.Videos
-
Breaking News | cGMP Manufacture | Drug Development | Industry News

Contract Pharma Q&A with Rich Loughlin of CMIC
CMO industry veteran discusses pharmaceutical outsourcing trends.Videos
-

David Tyler of CPL Sits Down with Contract Pharma
Director of Business Development discusses outsourcing trends and opportunities.10.17.22
-
cGMP Manufacture | Drug Development | Solid Dosage/Creams/Ointments

Contract Pharma Q&A with Drew Bradley of Adare Pharma Solutions
CDMO business development VP discusses pharmaceutical outsourcing trends.Videos
-
cGMP Manufacture | Drug Development | Fill/Finish | R&D

Contract Pharma Sits Down with Patrick Haffey of Selkirk Pharma
CEO of Fill/Finish CMO shares insight on the future of the Pharmaceutical industry.Videos
-
Bio News | Breaking News | Collaborations & Alliances | Industry News

Contract Pharma’s 2021 Salary Survey is Here!
Win one of three $100 Amazon gift cards!Tim Wright, Editor, Contract Pharma 05.14.21
-
-
-
Bio News | Breaking News | Collaborations & Alliances | Industry News | Packaging & Tracking | Promotions & Moves

Contract Pharma Audience Survey
Take a minute to answer a few questions and you could win a $100 Visa gift card!Contract Pharma Staff 11.06.20
-
Clinical Trials | CRO News | Drug Development | Information Technology | R&D

CRO Industry Update
Serge Bodart of IDDI provides insight on the pharma and biopharma trends impacting CROs, outsourcing in today’s market, and advances in clinical research.Online Exclusives Kristin Brooks, Contract Pharma 04.27.20
-
Breaking News | Industry News

CPL Launches Production of Hand-Sanitizer to Meet Critical Shortages
The first approved product is a hand sanitizer solution containing 80% alcohol in 60mL and 200mL bottlesContract Pharma Staff 04.21.20
-
Breaking News | Industry News
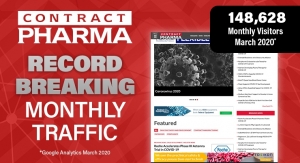
Contract Pharma Highlights Record Breaking Month
COVID-19 Industry Monitor propels ContractPharma.com to record breaking web traffic in March 2020Contract Pharma Staff 04.09.20
-
Clinical Trials | Information Technology | QA/QC

Managing Clinical Trials Amid the Coronavirus Pandemic
Patrick Hughes of CluePoints discusses the impact on clinical trials and how leveraging data may herald a long overdue paradigm shift in the industry.Online Exclusives Kristin Brooks, Contract Pharma 04.06.20
-
Biologics, Proteins, Vaccines | cGMP Manufacture | Clinical Trials | Drug Development | Scale-up/Technology Transfer

Rapid Delivery of Medical Countermeasures Amid Pandemic
Robert Erwin of iBio discusses the company’s COVID-19 vaccine development efforts and FastPharming manufacturing platform for the rapid delivery of vaccinesOnline Exclusives Kristin Brooks, Contract Pharma 04.03.20
-
Biologics, Proteins, Vaccines | Clinical Trials | Drug Development

COVID-19 Vaccine Response
ICON execs discuss developments in vaccine research efforts and key challenges of delivering a coronavirus vaccineOnline Exclusives Kristin Brooks, Contract Pharma 04.01.20
-

COVID-19 & Fear of the Unkown
We should be just as concerned with curbing the spread of fear, as we are the coronavirusTim Wright, Editor, Contract Pharma 04.01.20
-
Aseptic Processing | Facilities | Fill/Finish

GRAM Continues Aggressive Expansion in Parenterals
Backed by Arlington Capital Partners, Grand River Aseptic Manufacturing invests in major expansion of capacity and capabilities with new fill finish facilityTim Wright, Editor, Contract Pharma 04.01.20
-
Drug Delivery

CEO Spotlight: Will Downie
Vectura’s chief executive talks trends in the inhaled-drug market, and the company’s transition into a CDMOTim Wright, Editor, Contract Pharma 04.01.20
-
Coronavirus Crisis: Industry Impact Survey
We polled our readers to find out how you and your colleagues are being impacted by the coronavirus pandemic?Tim Wright, Editor, Contract Pharma 04.01.20
-
Coronavirus SARS-CoV-2: Architecture of a Pandemic
Deconstructing the epidemiology of a global panic.Ben Locwin, Contributing Editor 04.01.20
-
cGMP Manufacture | Drug Development | Logistics | Supply Chain
COVID-19: Business Update
Thermo Fisher Scientific provides perspective on the COVID-19 pandemic and how the company is working to help minimize disruptionsOnline Exclusives Kristin Brooks, Contract Pharma 03.30.20
-
Biologics, Proteins, Vaccines | cGMP Manufacture | Clinical Trial Materials | Drug Development

Newsmakers: Thermo Fisher Scientific
Leon Wyszkowski discusses the company’s recent investments, the challenges biopharma companies face, and future needs in the outsourcing spaceOnline Exclusives03.16.20
-

2020 Contract Manufacturing Survey
Now in its third year, our audience poll offers insights on why brands outsource manufacturing and what they look for.Sean Moloughney, Editor, Nutraceuticals World 03.04.20
-

Bora: Fast-growing CDMO Expands in U.S.
Contract Pharma talks with Samuel Ricchezza about the Taiwan-based CDMO’s push into North America.Tim Wright, Editor, Contract Pharma 03.04.20
-

Pharma Packaging Roundtable
Contract Pharma talks trends with contract packagers.Tim Wright, Editor, Contract Pharma 03.04.20
-
Bioanalytical Services | Breaking News | cGMP Manufacture | Clinical Trial Materials | Clinical Trials | Industry News

Altasciences Acquires Alliance Contract Pharma
Adds contract manufacturing and analytical services to early phase research offering02.27.20
-
Clinical Trials | Drug Development | R&D

Rare Disease R&D
Finding the hard-to-reach and leveraging big data and machine learning in drug developmentOnline Exclusives Kristin Brooks, Contract Pharma 02.25.20
-
Bio News | Biologics, Proteins, Vaccines | cGMP Manufacture

Newsmakers: Vigene Biosciences
Dr. Jeffrey Hung, Chief Commercial Officer, discusses Vigene’s new $20 million lab and strategic partnership with ASC TherapeuticsOnline Exclusives Kristin Brooks, Contract Pharma 02.20.20
-
Clinical Trials | Drug Development

Three Ways Clinical Trials Will be Enhanced in 2020
Insights on the major changes shaping the clinical landscapeRichard Young, Henry Levy, Jim Reily, Veeva Systems 02.13.20
-
cGMP Manufacture | Information Technology | Scale-up/Technology Transfer

The Future of Pharmaceutical Manufacturing
Neo PLM’s Cathal Strain discusses the need for a digital revolution to drive manufacturing innovationOnline Exclusives Kristin Brooks, Contract Pharma 02.12.20
-
Facilities

CEO Spotlight: Vincent Dunne
Wasdell Group's chief executive talks trends and plans for continued growthOnline Exclusives Tim Wright, Contract Pharma 02.10.20
-
Breaking News | Clinical Trial Materials | Clinical Trials | Industry News

Almac Launches Enhanced LAS
Aims to deliver an efficient, seamless process for clinical labels from protocol to print02.06.20
-
Biologics, Proteins, Vaccines | Clinical Trials | Drug Development | Industry News

Outsourcing in the Cell & Gene Therapy Space
Specialized and coordinated capabilities aim to accelerate drug development and reduce risk for sponsorsOnline Exclusives Kristin Brooks, Contract Pharma 02.04.20
-
Breaking News | Clinical Trial Materials | Clinical Trials | Industry News

Almac, Pfizer Report Success of Gene Therapy Trial
LEAN packaging process supports Pfizer’s mission to dose a child with life enhancing gene therapy01.31.20












