Search Results for 'isolation'
Contract Service Directory Companies
Solution Center Companies
View All Buyers Guide Companies »
-
Breaking News | cGMP Manufacture | Collaborations & Alliances

Multiply Labs and GenScript Partner to Automate Cell Isolation
Will introduce a fully automated solution for bead-based cell isolation.04.25.24
-
Biologics, Proteins, Vaccines

Isolation and Characterization of Product-Related Variants in Biotherapeutics
Biotherapeutics comprise a diverse class of protein-based drugs with intricate structural and functional characteristics.
-
Coronavirus SARS-CoV-2: Architecture of a Pandemic
Deconstructing the epidemiology of a global panic.Ben Locwin, Contributing Editor 04.01.20
-

-
-
Bio News | Bioanalytical Services | Biologics, Proteins, Vaccines | Breaking News | Clinical Trial Materials | Industry News

Arranta Bio Acquires Captozyme
Creates a CoE for microbiome development and clinical supply.11.13.19
-

Concentration and Reformulation of Cellular Immunotherapies – A Major Downstream Processing Step
...
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Collaborations & Alliances | Industry News

Thermo Fisher Teams up with CDMO Arranta Bio
Arranta also completes $82 million funding round.10.30.19
-
cGMP Manufacture

Just in Time Manufacturing
Changing perceptions of Just in Time Manufacturing in a clinical supply environment.Natalie Balanovsky, Just in Time Manufacturing Solutions Manager, Almac Clinical Services 10.16.19
-
Cytotoxics and High Potency Manufacturing

Handling & Assessing HPAPIs
As oncology pipelines grow, so too is the demand for highly potent materials, which require careful handling and containment.Jason Korbel, Technical Services Manager, Cambrex Charles City 09.16.19
-
Industry News

Gemini Bioproducts, Nordmark Biochemicals Enter Distribution Pact
Nordmark’s line of collagenase and neutral protease products can be purchased directly from Gemini08.09.19
-
Collaborations & Alliances | Drug Development
Sinovant and Roivant Sciences Launch Cytovant Sciences
New company focused on developing and commercializing innovative cellular therapeutics in Asia04.04.19
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Drug Development | Industry News

Orgenesis Signs Master Service and JV Agreement
Partners with TheraCell Advanced Biotechnology for the development of cell and gene therapies03.29.19
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Industry News

Lonza Establishes Custom Cell Biology Services
To offer unique manufacturing and testing capabilities03.29.19
-
Bio News | Drug Development | Facilities | Industry News | Promotions & Moves | R&D

HemaCare Moves Into New HQ
Company has officially moved its global headquarters to Northridge, California, in a 40,000 sq. ft. building in the heart of the San Fernando ValleyVideos
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Collaborations & Alliances | Industry News

Selexis, Berkeley Lights Form Cell Line Collaboration
Effort designed to speed drug candidates into the clinic10.18.18
-
Analytical Services | Bio News | Collaborations & Alliances | Drug Discovery | Industry News | Information Technology | Laboratory Testing | R&D
FloDesign, Cognate Announce Collaboration
To advance the development of life saving therapeutics by improving manufacturing efficiencies using FloDesign's ACP Platform09.12.18
-
APIs | Cytotoxics and High Potency Manufacturing

HPAPI Technology Evolution
Handling of high potency compounds for API and DP from development on a laboratory scale and scale-up to commercial.Martina Kotthaus, Jeff Dinyer, Anil Kane and Marvin Faber, Patheon, part of Thermo Fisher Scientific 09.11.18
-
cGMP Manufacture | Solid Dosage/Creams/Ointments

Continuous Pharmaceutical Manufacturing
Exploring the advantages of continuous over batch manufactureShawn Conway, Ph.D. and Daniel Bowles, Ph.D., Cambrex 09.11.18
-
Analytical Services | Bio News | Drug Discovery | Industry News | Laboratory Testing
SGS Implements PBMC Analysis
For the collection & use of peripheral blood mononuclear cells (PBMCs) for early phase clinical trials at its CPU in Belgium09.05.18
-
Cold Chain Management | Logistics
The Chain of Compliance
Why the Chain of Compliance is critical to winning in the RMAT marketMark W. Sawicki, Chief Commercial Officer, Cryoport 06.12.18
-
Bio News | Bioanalytical Services | Breaking News | Facilities | Industry News

Saint-Gobain Opens Life Sciences Lab in Mass.
New lab is dedicated to application testing of single-use systems for the cell and gene therapy market05.14.18
-
Biologics, Proteins, Vaccines
-
Bio News | Breaking News | cGMP Manufacture | Facilities | Industry News

Quay Pharma Launches New GMP Suites
As part of an investment in the company's live biotherapeutics services04.04.18
-
Bio News | cGMP Manufacture | Drug Delivery | Facilities | Industry News | Laboratory Testing | Risk Management

Quay Expands Specialist Isolation Suites
The suites are to support the development of potent drugs in a protective environment03.06.18
-
Laboratory Testing | Reader Showcase
Thermo Fisher Launches Lab Automation Solutions
inSPIRE vertical modular robotic platform designed to integrate multiple instruments in a space-saving solution03.01.18
-

Arcinova Strengthens API Services with New GMP Mfg. Capabilities
Multi-service contract research and development organization makes major investment11.10.17
-
Sterile Technology Trends
Exploring the benefits of sterile transfer valve technologyChristian Dunne, ChargePoint Technology 10.11.17
-
Drug Development | Industry News | Trials & Filings

-
Analytical Services | Chemistry | Industry News | Laboratory Testing | Methods Development

Avista Pharma Completes Durham HQ Expansion
Analytical Research and Development and Quality Control labs have doubled in size and expanded service offerings08.08.17
-
APIs

Optimizing the API Life Cycle Through Innovation
Bringing down API costs through new technologies and customer collaborationJonathan Knight, VP New Product Development, Cambrex 06.06.17
-
Drug Development | Drug Discovery
Tackling TB Resistance
The TB Alliance is partnering with universities, government agencies, public agencies and a small number of pharma companiAdele Graham-King, Contributing Editor 05.09.17
-
Biologics, Proteins, Vaccines
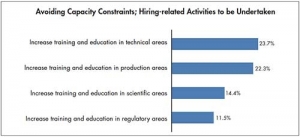
CMOs Hiring Constraints: Cell Therapy Manufacturing Expecting Future Challenges
Technically trained staff needed to run complex manufacturing platforms, automationEric S. Langer, BioPlan Associates 03.07.17
-

Risks to Assess When Selecting Clean Benches And Biosafety Cabinets For Animal Research
Acumen White Papers - Volume 3 No. 1
-
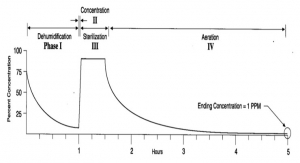
Cycle Parameters For Decontaminating A Biological Safety Cabinet Using H2O2 Vapor
Acumen White Papers - Volume 1 No. 2
-
Analytical Services | APIs | Breaking News | Chemistry | Collaborations & Alliances | CRO News | Drug Development | Industry News | Validation
-
Drug Development | Drug Discovery
How ‘Mush-room’ For Improvement in Antibiotic Production?
The medical world is acutely aware of the issues on the horizon with regards to antibiotic resistanceAdele Graham-King, Contributing Editor 06.02.16
-
Chemistry | Clinical Trials | Drug Discovery | Industry News | Laboratory Testing | Preclinical Outsourcing
Novasep Opens Expanded U.S. Kilo Lab
Begins production of batches of synthetic molecules for early stage development06.02.16
-
Breaking News | Industry News | Reader Showcase
MilliporeSigma Expands Line of Guava Flow Cytometers
Optical configurations meet needs of lab, research discipline or individual application05.11.16
-
Drug Delivery | Drug Development | Excipients | Process Development | Process Validation | Risk Management | Validation

Process Development Trends
An approach to process development of pMDIs using cold fill and pressure fill technologySteve Haswell, 3M Drug Delivery Systems 05.05.16
-
Capsules | Clinical Trials | Excipients | Formulation Development | Serialization

The Evolution of the ‘One-Stop Shop’ in Clinical Supplies Sourcing
What does a one-stop shop really mean and is it always best for the client?Paul Skultety, Alex McClung, Ted Koontz and Damian Gant, Xcelience, a division of Capsugel Dosage Form Solutions 05.05.16
-
Excipients | Industry News
Reliable Biopharma Expands Manufacturing Capacity
Increases output of lithium dodecyl sulfate and sodium dodecyl dulfate in its St. Louis facility02.22.16
-
APIs | cGMP Manufacture | Excipients
AAI/CML Expands Mfg. Capabilities in The Netherlands
Adds DE Dietrich Agitated Filter Dryer12.22.15
-
-
-
-
-












