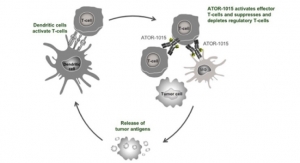
Bavarian Nordic announced a new collaboration that will investigate CV301, the Company's targeted immunotherapy candidate, and durvalumab (IMFINZITM), AstraZeneca's PD-L1 inhibitor, in combination with maintenance chemotherapy for patients with metastatic colorectal or pancreatic cancers.
Under the collaboration with Georgetown University, both Bavarian Nordic and AstraZeneca will contribute clinical trial material and provide financial support for a planned Phase 1/2 clinical study. The trial, which is being sponsored by Georgetown University, will be led by Dr. Michael Pishvaian, Assistant Professor in the Department of Hematology/Oncology at the Lombardi Comprehensive Cancer Center at Georgetown University Medical Center.
The Phase 1/2 trial will begin with a lead-in study to determine the safety and tolerability of the combination, as well as the recommended Phase 2 dose of durvalumab in combination with CV301 and chemotherapy. The Phase 2 portion of the study will consist of two parallel trials, enrolling up to 26 patients for each disease setting. The primary endpoint for both arms of the study will be progression-free survival (PFS) with multiple secondary endpoints, including objective response rate (ORR), overall survival (OS), and disease control rate (DCR).
"With this trial, we are hopeful to continue demonstrating CV301's potential in multiple cancers and combinations, particularly in a treatment setting in which checkpoint inhibition alone has yet to show significant benefit. We are very grateful to both the investigators and AstraZeneca for their support, and look forward to learning more as the data becomes available," said Paul Chaplin, president and chief executive officer of Bavarian Nordic.