Search Results for 'Stability'
-
Aseptic Processing | Biologics, Proteins, Vaccines | Fill/Finish | Lyophilization | Parenterals
Biopharma Fill Finish Contract Manufacturing Market
Outsourcing strategies and areas of market opportunitiesCindy Liu and William Downey, HighTech Business Decisions 04.20.17
-
Bio News | Bioanalytical Services | Bioassay Development | Biologics, Proteins, Vaccines | Breaking News | Clinical Trial Materials | Clinical Trials | Collaborations & Alliances | Industry News
Cobra Biologics Helps Alligator Bio Complete Phase I Trial
maxXpress platform used to enable rapid and high-yielding expression of protein ADC-101304.12.17
-
Bio News | Bioanalytical Services | Breaking News | Facilities | Industry News

Eurofins Expands Biopharma Testing Operations
New £4 million UK facility to house biopharmaceutical testing business04.06.17
-
Clinical Trials | Drug Development | Formulation Development | Industry News

Ligand Licenses Captisol-Enabled Ganaxolone
Marinus has exclusive worldwide rights to Captisol-enabled ganaxolone for use in humans04.05.17
-
Analytical Services | Cytotoxics and High Potency Manufacturing | Drug Development | Industry News
Alligator Bioscience Trial Employs Cobra’s MaxXpress Platform
Develops cell line expressing ADC-1013 for CD40 agonistic immuno-oncology using maxXpress service platform04.03.17
-
Elemental Impurities: A Virtual Company Perspective
A look at risk assessments for elemental impurity determinations in oral tablet and parenteral drug products.Anthony DeStefano and Thomas Kester, Recordati Rare Diseases, Inc. 04.03.17
-
Analytical Services | Industry News

-
Breaking News | Industry News
Avista Pharma Spends $4mn to Upgrade Capabilities
Expands microbiology, sterility testing and microbial ID capabilities at Agawam facility03.27.17
-
Drug Development | Drug Discovery
The Trials and Tribulations of Male Contraception
Where has research led in the search for effective oral male contraception?Adele Graham-King, Contributing Editor 03.07.17
-
Solid Dosage/Semi-solids

Oral Solid Dosage Outsourcing: Trends & Challenges
Despite the increasing number of biologics, oral solids remain the dominant dosage form in the pharmaceutical market.Tim Wright, Editor, Contract Pharma 03.07.17
-
Chemistry | Drug Development
Know Your Drug: A Solution to Dissolution
Examining key strategies for solubility and dissolution assessment of new drug products, drug product intermediates and APIsLuís Sousa, Mafalda Paiva and Pedro Serôdio, Hovione 03.07.17
-
Solubility Enhancement: Novel Approaches to Rational Formulation Choice
Novel approaches to rational formulation choiceSanjay Konagurthu, Tom Reynolds and Matt Wessel, Patheon 03.07.17
-
Drug Development | Formulation Development
Modeling & Simulation for Drug Development & Formulation
A look at how to leverage modeling and simulation technology to enhance all phases of the drug development process.Nathan Teuscher and Nikunjkumar Patel , Certara 03.07.17
-
Analytical Services | Breaking News | Collaborations & Alliances | Drug Development | Drug Discovery | Industry News
Alcami Expands Analytical Capabilities
Invests in Bruker's D8 Discover HTS System for high-throughput X-Ray diffraction screening03.06.17
-
Breaking News | Industry News
MilliporeSigma Offers Fully Integrated ADC Services
Designed to reduce complexity, accelerate time to market02.23.17
-
Serialization | Supply Chain

Packaging Equipment Technology Preview
Today’s complex drug products present challenges; the latest equipment capabilities help overcome themOnline Exclusives02.15.17
-
Breaking News | GMPs/GCPs | Industry News
PPD Expands Stability Storage and Sample Distribution
Increase in capacity and capabilities at Wisconsin GMP lab designed to support growing client needs12.14.16
-
Biologics candidate enters clinical trials for first time using CORYNEX® Protein Expression System
...
-
Industry News
Metrics Invests in Stability Storage Expansion
Will triple current capacity at Greenville campus08.24.16
-
Breaking News | Collaborations & Alliances | Industry News
Catalent Biologics and Zumutor Team Up
Collaborate to develop antibodies with enhanced ADCC activity08.03.16
-
Formulation Development | Validation
Altimmune Receives $120M BARDA Contract
Funds support research for clinical development of first-in-class anthrax vaccine candidate NasoShield08.01.16
-

-
"Bilcare Academy" Event Sept. 21 at The Hyatt Regency New Brunswick
Will cover a range of topics dealing with the crucial role that packaging plays in the pharmaceutical life cycle07.05.16
-
Biosimilars | Breaking News | CRO News | Drug Development | Industry News | Validation
Charles River Acquires Blue Stream Labs
Offers clients an integrated portfolio for biologic and biosimilar drug development06.28.16
-
Drug Development | Excipients | Lyophilization | Risk Management

Clinical Manufacturing On Time and Within Budget
...David Brett, Vetter Pharma International GmbH 06.02.16
-
Biosimilars | Drug Delivery | Injectables | Parenterals | Serialization | Supply Chain | Vials

Contract Packaging Market Trends
Contract packagers talk about hot growth areas and the looming 2017 DSCSA deadlineTim Wright, Editor 06.02.16
-
Breaking News | Chemistry | CRO News | Microbiology | Validation
-
-
Bio News | Breaking News | Industry News
Ajinomoto Althea Receives Patent for Manufacturing Crystal Monoclonal Antibodies
Methods aid stabilization, storage and delivery of biologically active antibody crystals05.17.16
-
APIs | Breaking News | Industry News
Capsugel Expands Micro-Dosing Services
Combines proprietary Xcelodose technology with best practices established at Xcelience05.17.16
-
Analytical Services | cGMP Manufacture | GMPs/GCPs | Industry News | Inspections
WuXi AppTec’s Shanghai Facilities Pass EMA Inspection
Completes third successful EMA inspection for GMP compliance05.12.16
-
Bio News | Biologics, Proteins, Vaccines | Breaking News | Facilities | Industry News | Process Development | R&D
Fujifilm Diosynth Opens New Lab
Construction now complete on new 62,000 square foot facility in North Carolina05.11.16
-
Drug Delivery | Drug Development | Excipients | Process Development | Process Validation | Risk Management | Validation

Process Development Trends
An approach to process development of pMDIs using cold fill and pressure fill technologySteve Haswell, 3M Drug Delivery Systems 05.05.16
-
Biosimilars | Clinical Trials | Formulation Development
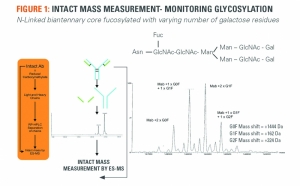
Critical Steps for Biosimilar Assessment
Establishing “finger-print like” biosimilarity prior to the clinicFiona Greer, SGS 05.05.16
-
Capsules | Clinical Trials | Excipients | Formulation Development | Serialization

The Evolution of the ‘One-Stop Shop’ in Clinical Supplies Sourcing
What does a one-stop shop really mean and is it always best for the client?Paul Skultety, Alex McClung, Ted Koontz and Damian Gant, Xcelience, a division of Capsugel Dosage Form Solutions 05.05.16
-
Analytical Services | GMPs/GCPs | Industry News | Inspections | Laboratory Testing
-
Logistics | Validation

CMO Systems
A CMO system includes four aspects of the product lifecycle: regulatory, technical, compliance, and operations.Philip K. Burns, CRO Consulting 04.05.16
-
Biosimilars | Process Development

Biopharmaceutical Contract Manufacturing Contract Negotiations
Navigating the complexities of supply agreements.William Downey, HighTech Business Decisions 04.05.16
-
Capsules | cGMP Manufacture | Clinical Trials | Drug Development | Drug Discovery | Excipients | Facilities | Formulation Development

Solid Dispersions
A universal formulation strategy for poorly soluble drugs?Robert Harris, Juniper Pharma Services 04.05.16
-
APIs | cGMP Manufacture | Drug Development | Facilities | Formulation Development | Supply Chain
-
Bioassay Development | Biosimilars | Breaking News | Collaborations & Alliances | Drug Discovery | Laboratory Testing | Validation
SGS, DiscoverX to Qualify and Supply Bioassays
Aims to provide commercial ready-to-assay cryopreserved cells in a qualified kit03.30.16
-
Breaking News | Industry News
Bilcare Research Invests in Aclar Laminates
Invests in a new laminator at its Wilmington, DE facility03.14.16
-
Analytical Services | Biosimilars
How to Build Better Proteins
Advancing the next generation of biologicsAileen Ruff, Catalent 03.09.16
-
Fill/Finish | Process Development | Validation
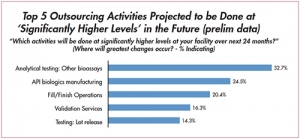
Taking Stock of Biopharma’s Outsourcing Markets
Outsourcing of testing services on the riseEric S. Langer, BioPlan Associates 03.09.16
-
Inspections | Risk Management | Supply Chain
Quality Metrics: What Does it Really Mean?
Background, thoughts and the current industry dialogue on FDA’s quality metrics initiativeDr. Mike Long, ValSource 03.09.16
-
APIs | Capsules | Clinical Trials | Drug Delivery | Excipients | Formulation Development

Developing Palatable Drug Products
A decision framework and guide for CDMOsJeff Worthington and David Tisi, Senopsys LLC 03.09.16
-
APIs | Capsules | Chemistry | Drug Delivery | Excipients | Process Development | Validation
Liquid Fill Hard Capsule Technology
A simple and versatile tool for new compounds and life cycle managementStephen Brown and Wei Tian, Encap Drug Delivery 03.09.16
-
Drug Delivery | Fill/Finish | Information Technology | Lyophilization | Serialization | Supply Chain | Vials

Packaging Equipment Close-Up
To meet the wide-ranging needs of the marketplace, suppliers and contract packagers are looking to deliver flexible solutions.Kristin Brooks, Contract Pharma 03.09.16
-
APIs | Biosimilars | Drug Delivery | Drug Development | Fill/Finish | Formulation Development | Lyophilization | Parenterals | Supply Chain

Parenteral Outsourcing Trends
Contract Pharma asked a roundtable of business leaders from Symbiosis, Grifols, Althea, LSNE and Baxter to discuss key trends in the parenteral dosage development and manufacturing marketTim Wright, Editor 03.09.16
-
APIs | Chemistry | Clinical Trials | Drug Delivery | Drug Development | Industry News | Injectables | Process Development

Consolidation in Outsourcing
Reviewing 2015’s M&A activityMichael A. Martorelli, Fairmount Partners 03.09.16












