Tim Wright, Editor06.02.15
The biopharmaceutical market continues to grow and more and more biopharma firms are calling upon the services of contract manufacturing organizations (CMOs), which as we all know offer various advantages for sponsors. Technical expertise, operational efficiency, regulatory support, the ability to allow sponsor companies to focus on their core competencies. How many times have we heard this drum being banged? However, the most valuable service CMOs probably offer is in serving as a cost-cutting tool, because let’s face it, at the end of the day business is all about boosting the bottom line. Right?
So CMOs have grown to be ubiquitous across the pharma and biopharma landscape. Are there even any pharma or biopharma firms left out there that don’t view outsourcing as a strategic imperative?
The biopharmaceutical contract manufacturing market has witnessed considerable growth in the past few years. There are estimated to be more than 160 biopharma CMOs operating in today’s market whose services range from cell line development and API manufacturing to fill/finish of the product.
Beginning on page 54, William Downey from High Tech Business Decisions takes a look at the trends in the current market, which he says is poised to reach $3 billion this year. Because of the greater demand for outsource services CMOs on the biopharma front are investing in new plant and equipment or acquiring capacity to meet future industry needs.
The strong growth in the biologics market is one of the major drivers pushing the biopharmaceutical contract manufacturing market forward in the coming years. By 2020 there is an estimated $67 billion worth of patents on biological products expiring. At the same time governments are under more and more pressure to reduce rising health care costs and biosimilars represent a major opportunity for the pharmaceutical industry.
However, understanding biosimilars from just about any perspective is not easy. Delving into the subject to try and sort through some of the confusion are PPD’s Joan Boren, Costantino Congiatu and Patricia Hurley. Beginning on page 58 the authors offer their perspective on the growing interest in biosimilars, which they say is evident by the approximately eightfold increase in the number of biosimilar clinical trials between 2007 and 2014. They also say the market growth of biosimilars is going to be significant and be worth approximately $2 billion by 2018.
Over the past ten years, regulatory authorities worldwide have been focusing on developing guidelines for biosimilars and this article addresses current regulations and challenges impacting the development of biosimilars.
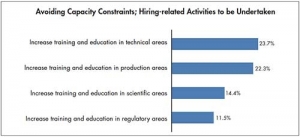
Also on the bio front, Eric Langer from Bioplan Associates and our resident BioPharm Insider (page 30), talks about the capacity constraints CMOs are facing, while in “Biosimilars and Their Future in Medicine” (page 22), our Clinically Speaking columnist Ben Locwin takes us through the first, simplest, but most important steps with biosimilars and that is understanding what they actually are! Hope you enjoy.
Tim Wright, Editor
twright@rodmanmedia.com
So CMOs have grown to be ubiquitous across the pharma and biopharma landscape. Are there even any pharma or biopharma firms left out there that don’t view outsourcing as a strategic imperative?
The biopharmaceutical contract manufacturing market has witnessed considerable growth in the past few years. There are estimated to be more than 160 biopharma CMOs operating in today’s market whose services range from cell line development and API manufacturing to fill/finish of the product.
Beginning on page 54, William Downey from High Tech Business Decisions takes a look at the trends in the current market, which he says is poised to reach $3 billion this year. Because of the greater demand for outsource services CMOs on the biopharma front are investing in new plant and equipment or acquiring capacity to meet future industry needs.
The strong growth in the biologics market is one of the major drivers pushing the biopharmaceutical contract manufacturing market forward in the coming years. By 2020 there is an estimated $67 billion worth of patents on biological products expiring. At the same time governments are under more and more pressure to reduce rising health care costs and biosimilars represent a major opportunity for the pharmaceutical industry.
However, understanding biosimilars from just about any perspective is not easy. Delving into the subject to try and sort through some of the confusion are PPD’s Joan Boren, Costantino Congiatu and Patricia Hurley. Beginning on page 58 the authors offer their perspective on the growing interest in biosimilars, which they say is evident by the approximately eightfold increase in the number of biosimilar clinical trials between 2007 and 2014. They also say the market growth of biosimilars is going to be significant and be worth approximately $2 billion by 2018.
Over the past ten years, regulatory authorities worldwide have been focusing on developing guidelines for biosimilars and this article addresses current regulations and challenges impacting the development of biosimilars.
Also on the bio front, Eric Langer from Bioplan Associates and our resident BioPharm Insider (page 30), talks about the capacity constraints CMOs are facing, while in “Biosimilars and Their Future in Medicine” (page 22), our Clinically Speaking columnist Ben Locwin takes us through the first, simplest, but most important steps with biosimilars and that is understanding what they actually are! Hope you enjoy.
Tim Wright, Editor
twright@rodmanmedia.com